The Organ You Were Told Doesn’t Matter After Childhood
Why thymus regeneration will define the next era of longevity science
In the fall of 2018, biomedical engineer Bobby Brooke gave a lecture to a small crowd at the Ending Age-Related Diseases conference in NYC. The topic of the talk was centered around the potential to regenerate the thymus as a way of reversing age-related immune decline.
Brooke opens up the presentation with a quote from a name that may look familiar.
Five years prior in 2013, Brooke co-founded Intervene Immune alongside Greg Fahy, a cryobiologist and the leading researcher of what’s become the now popular TRIIM Trial (Thymus Regeneration, Immunorestoration, and Insulin Mitigation).
As we await the results of this trial, the strongest evidence we’ve seen yet supporting the thymic-longevity thesis just landed in Nature.
What We Used to Believe
The way the medical textbooks are written makes it appear pretty straightforward. The thymus was a childhood organ. It matured T cells. It built the immune arsenal. Then it shrank into an inconsequential lump of fat by midlife.
It was the accepted view for decades. Until 2023, when researchers at Massachusetts General Hospital published a huge finding: adults who had their thymus surgically removed had significantly higher rates of death, cancer, & autoimmune disease.
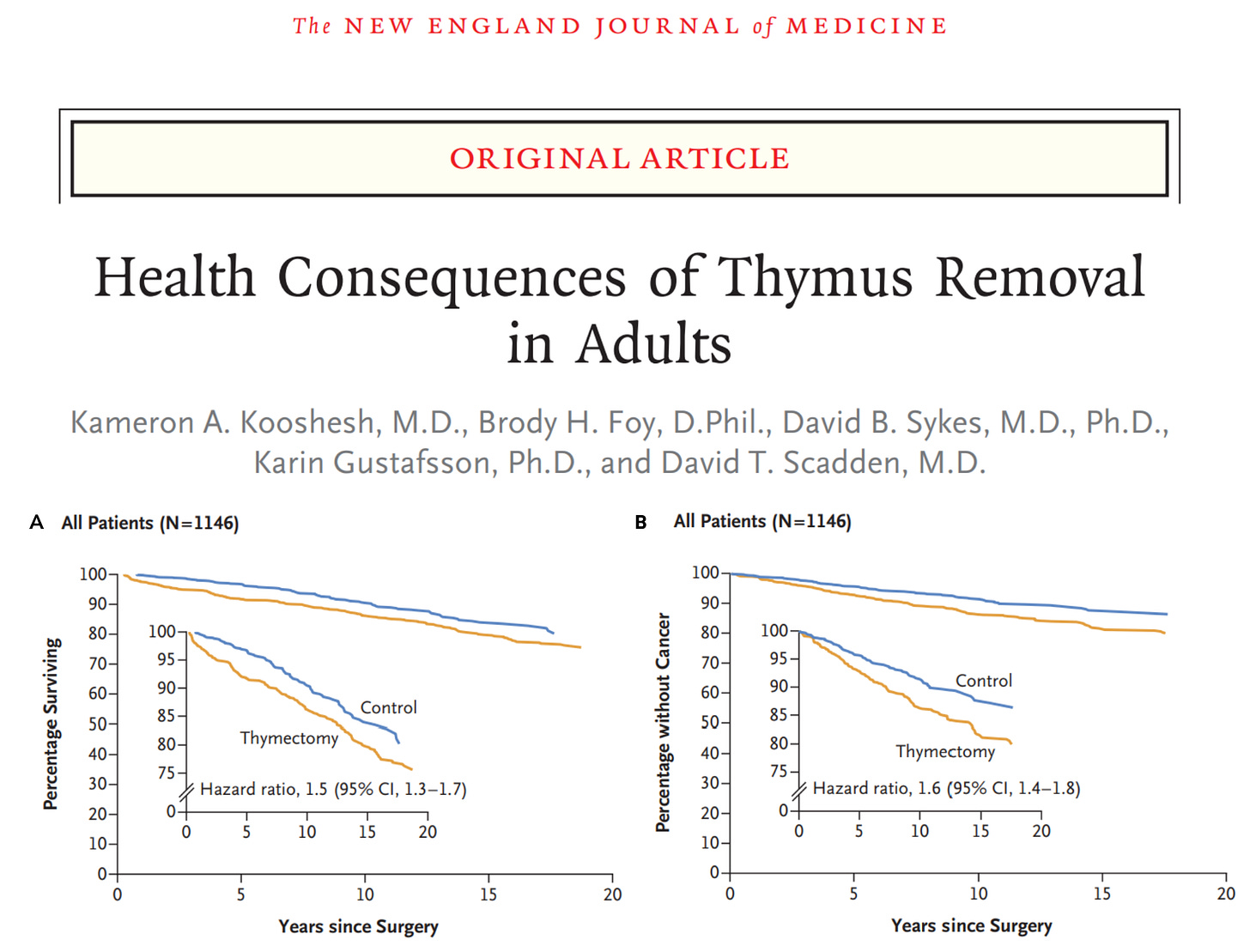
The Study
Researchers from Harvard, Maastricht University, & Aarhus University analyzed over 27k adults across two major prospective cohorts: the National Lung Screening Trial (NLST, n = 25,031) & the Framingham Heart Study (FHS, n = 2,581).
They built a deep learning system trained on thousands of CT scans to score “thymic health” quantifying how much functional thymic tissue remained versus fatty degeneration. Then they tracked outcomes over 12 years.
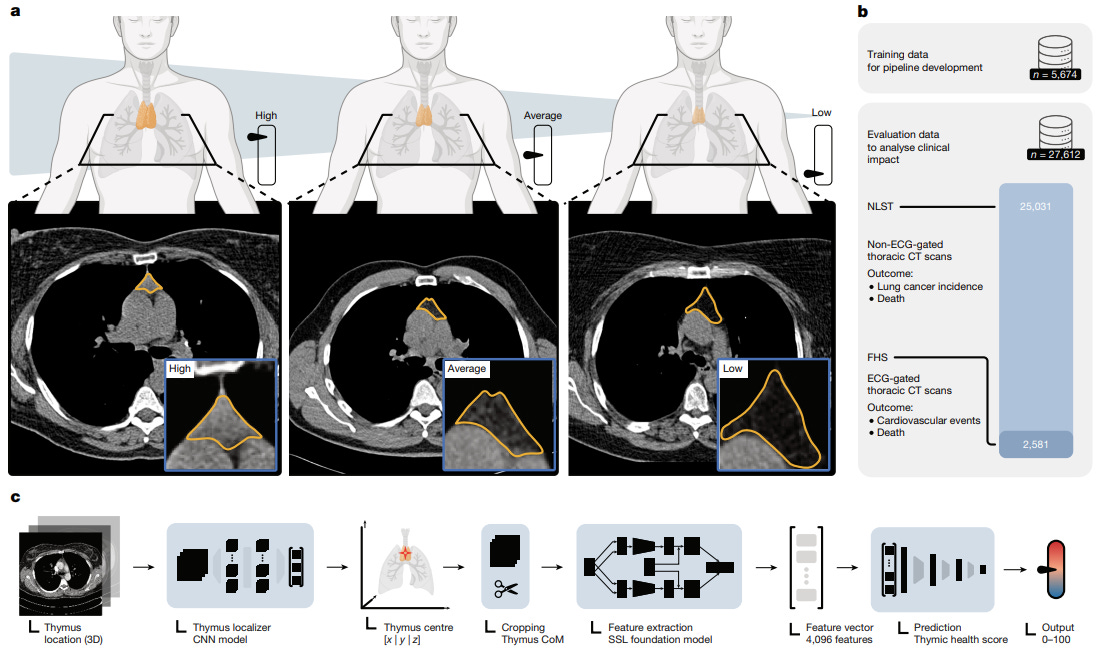
The Findings
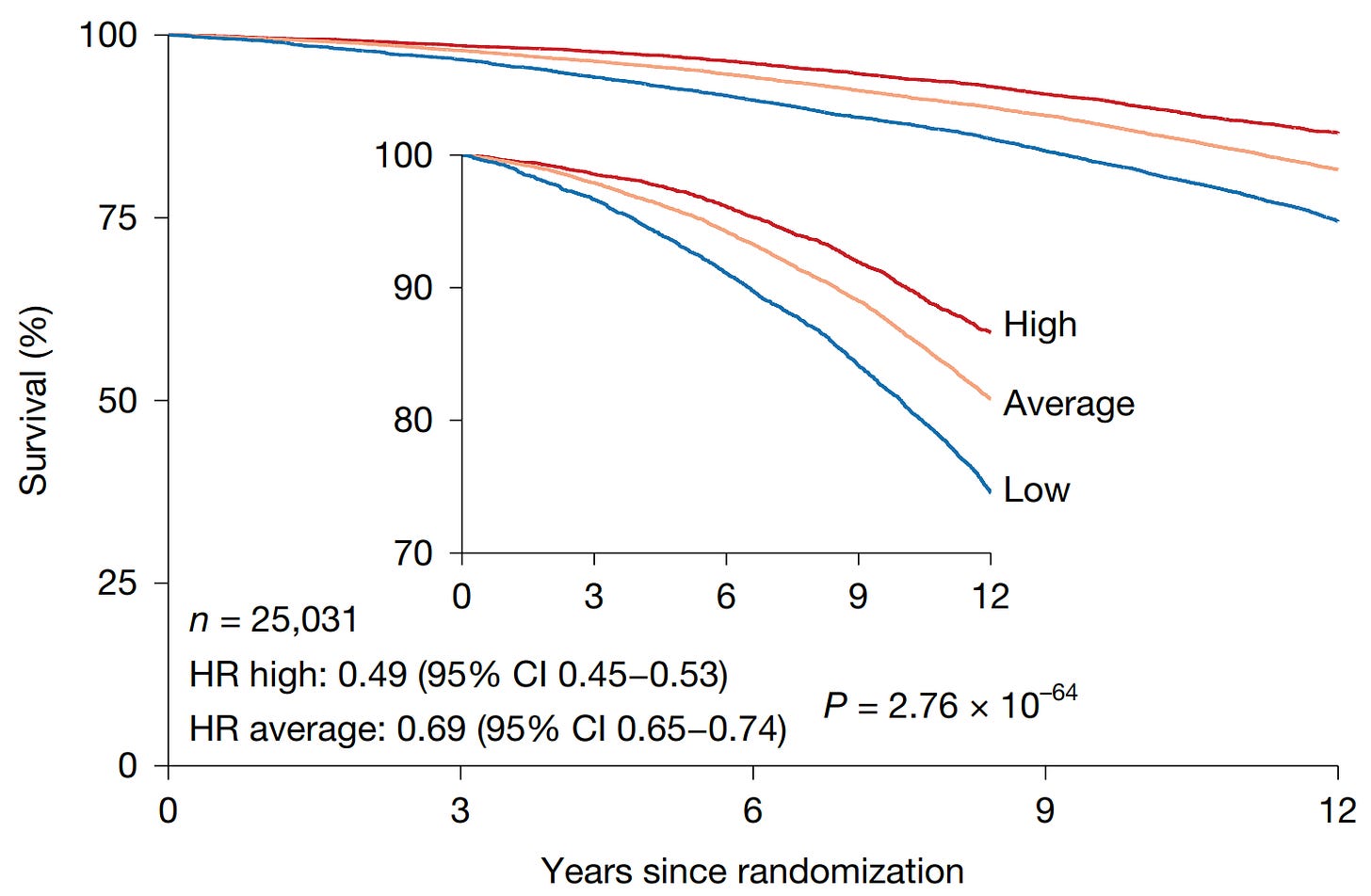
1. High thymic health = dramatically lower mortality.
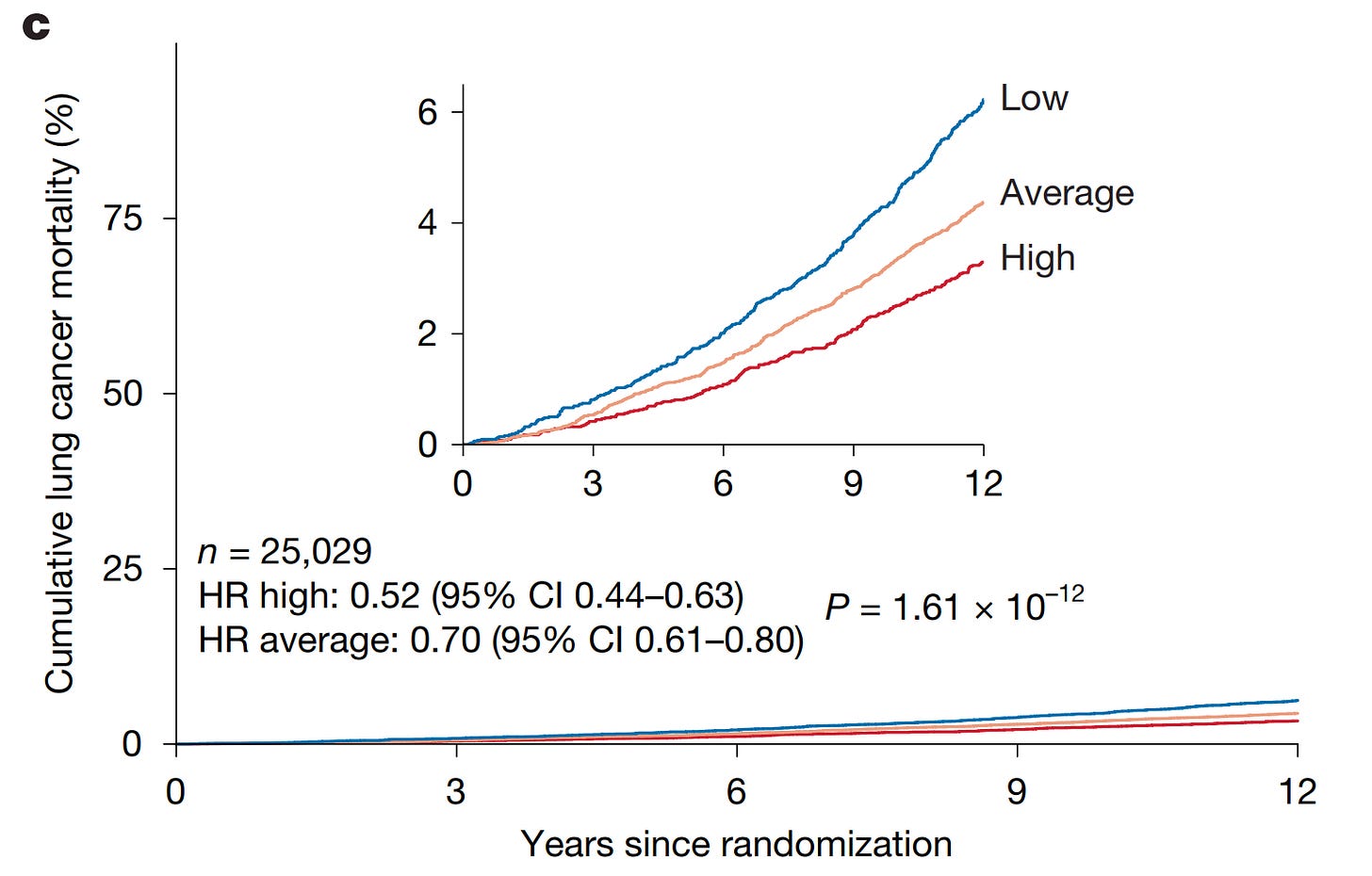
In the NLST, those with the highest thymic health scores had an ~50% reduction in risk of death from any cause compared to those with the lowest scores.
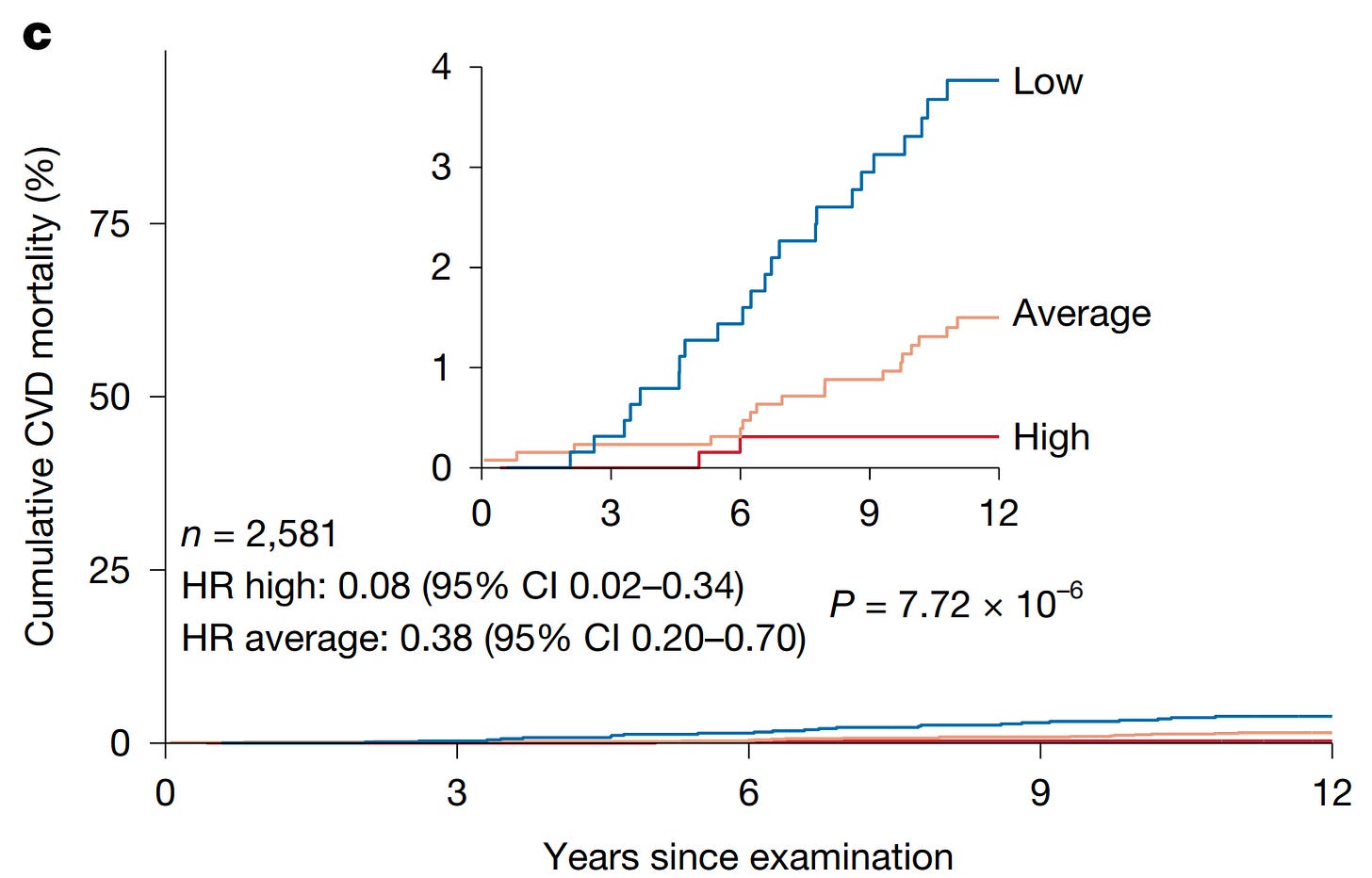
In the FHS, we get an even stronger effect. High thymic health was associated with a 76% lower risk of all-cause death.
Each step up in thymic health also corresponded to progressively lower mortality.
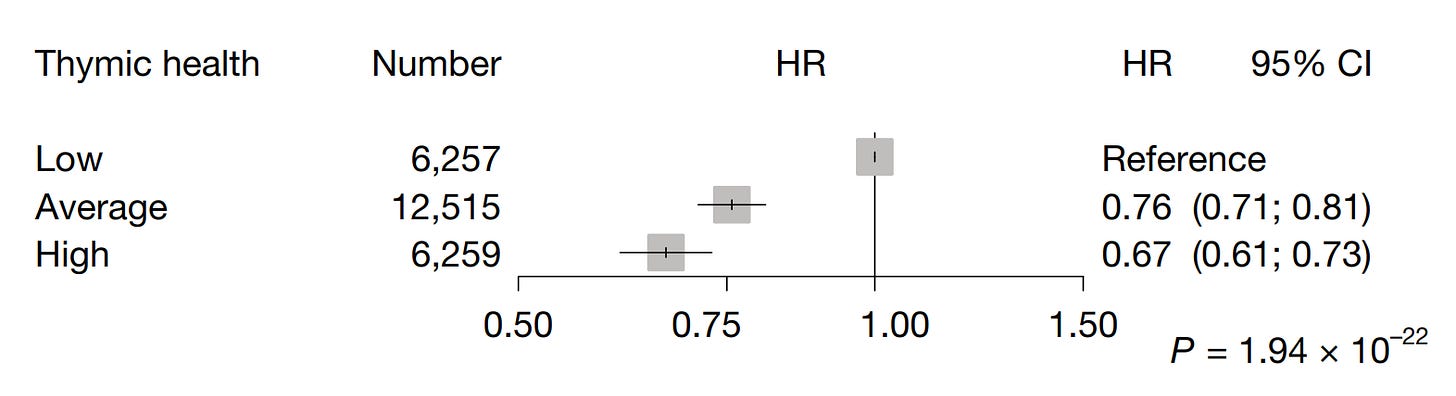
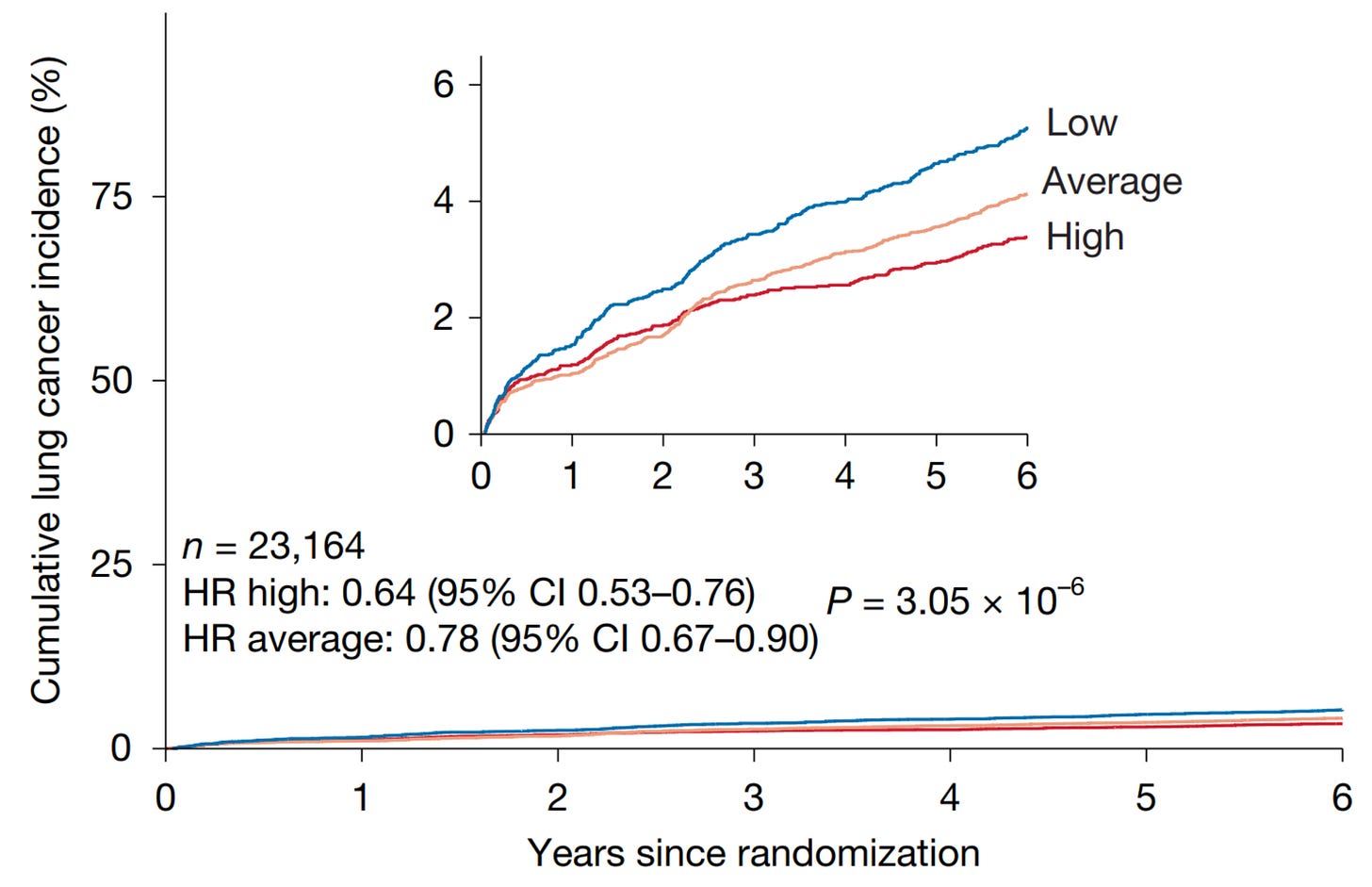
2. Lower cancer incidence and cancer-specific mortality.
Individuals with high thymic health were 36% less likely to develop lung cancer.
The high thymic health subjects also had ~50% less likely to die from it.
As the thymus involutes, two things happen simultaneously:
Naïve T cell output drops constricting the T cell receptor (TCR) arsenal. Fewer unique T cells = less defense to recognize novel tumor antigens. As we get older, age-related mutations produce tumor-specific neo-antigens and the naïve T cell pool fails to recognize them (Wang et al., 2020).
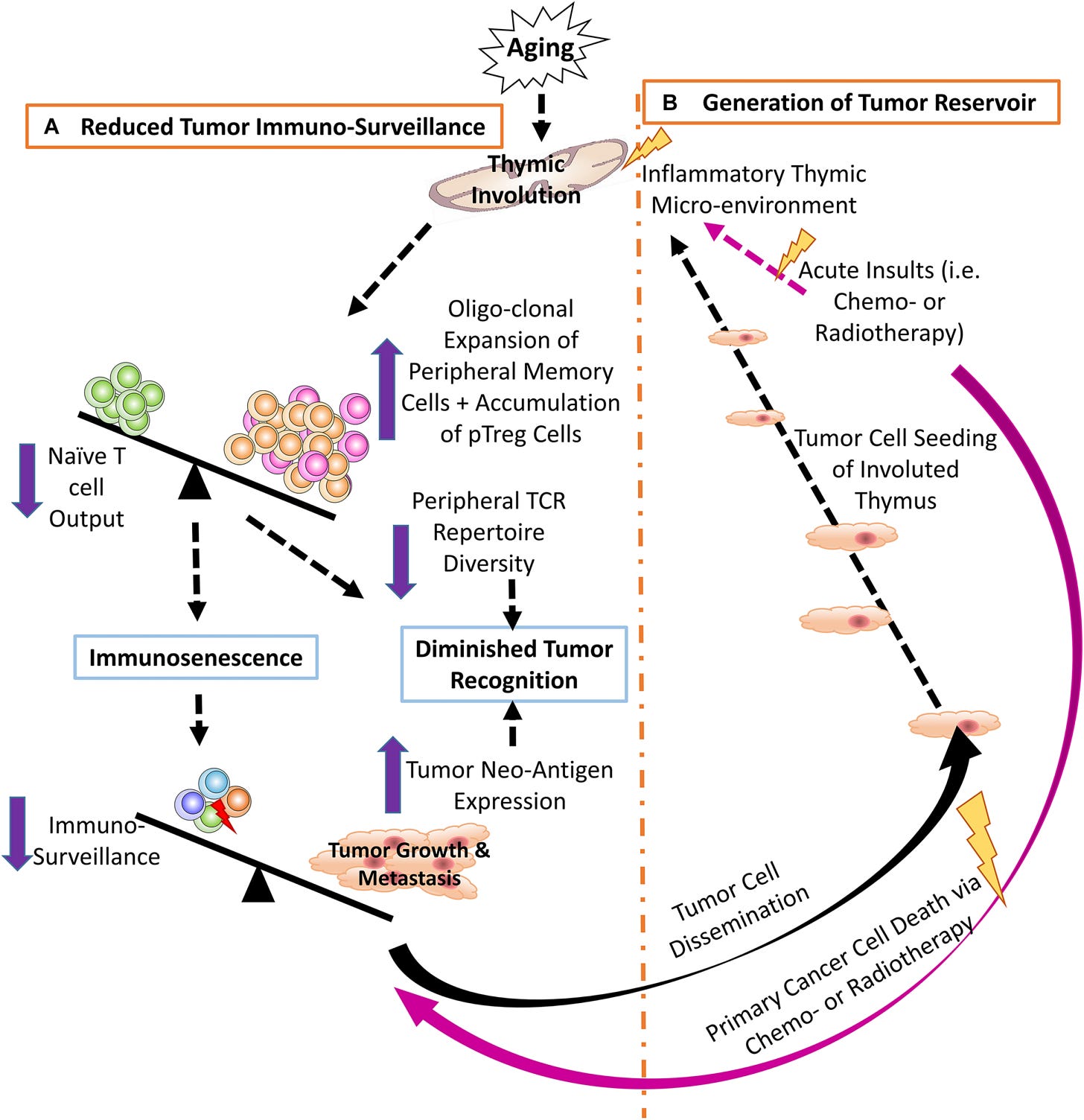
The involuted thymus shifts its output ratio toward regulatory T cells over effector T cells. More regulatory ones suppress anti-tumor immune responses. Senescent T cells upregulate PD-1 (programmed death 1) expression further enhancing the PD-1/PD-L1 axis that lets cancer cells evade destruction.
Palmer et al. (2018) predicted similarly: declining T cell output from thymic involution meant an age-related rise in cancer incidence. Across 25k+ people, they saw all forms of cancer mortality were significantly lower in the high thymic health group.
3. Cardiovascular disease risk dropped dramatically.
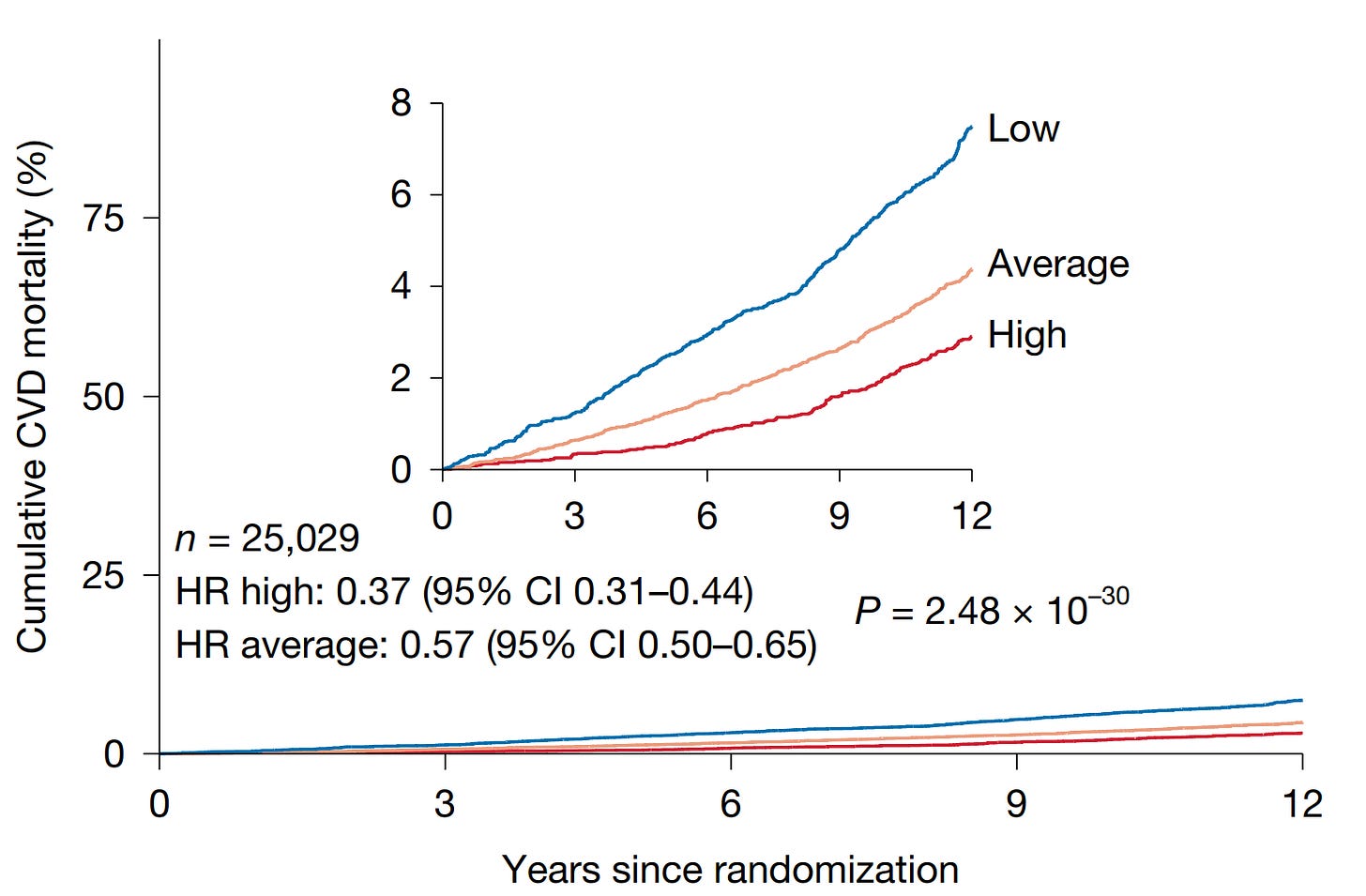
In the NLST, cardiovascular mortality was 63% lower in the high thymic health group.
In the FHS, it was 92% lower.
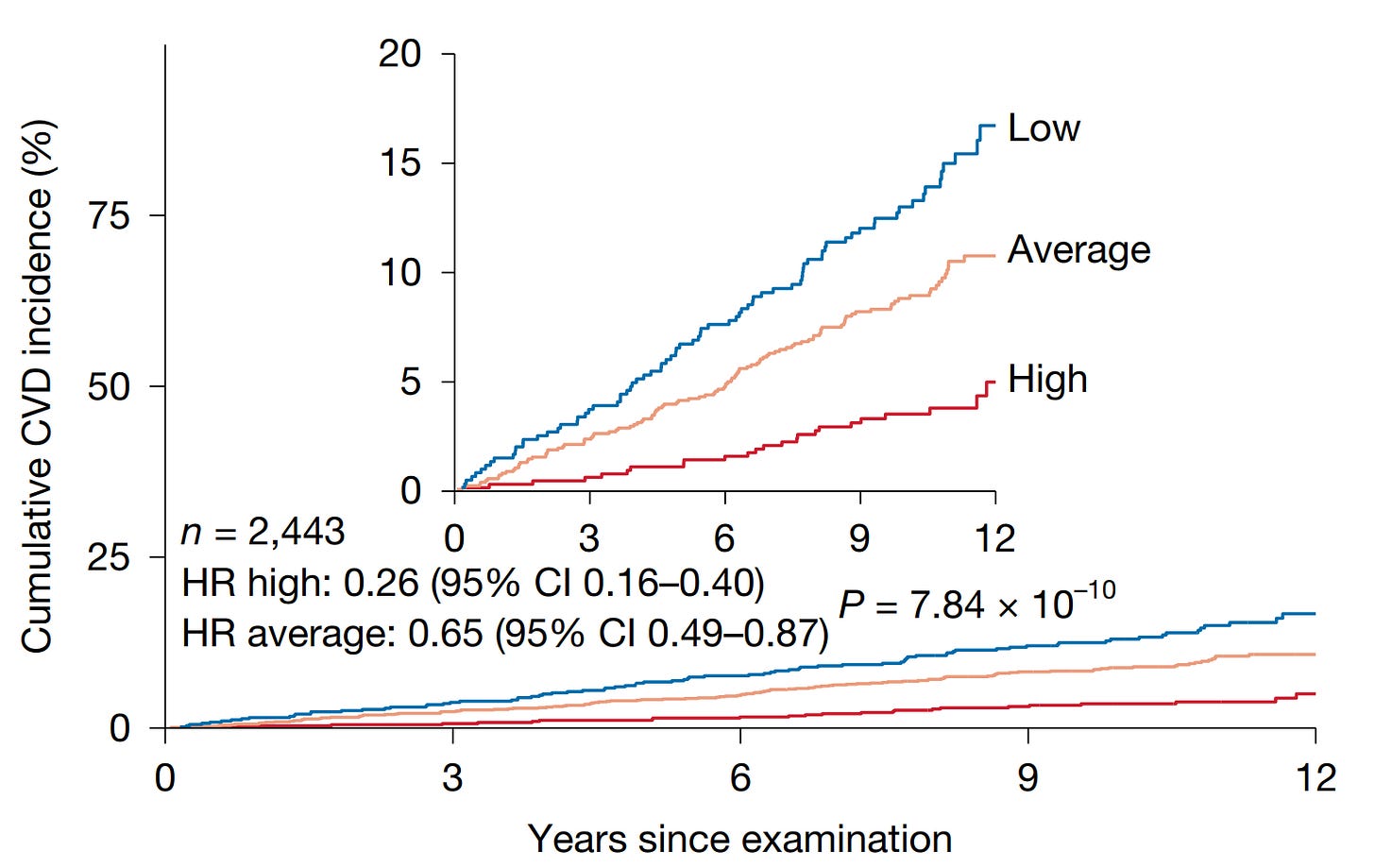
Subjects with high thymic health also had a 74% lower cumulative incidence of cardiovascular events like myocardial infarction, heart failure, & cerebral embolism.
Atherosclerosis is an immune-inflammatory disease so T cells are key to its progression:
Thymic output declines → the body compensates by cloning existing T cells rather than producing new ones → drives the accumulation of senescent T cells (shortened telomeres, restricted receptor diversity, & pro-inflammatory) → more pro-inflammatory cytokines → more destabilization of arterial plaques.
Thymic dysfunction also shifts the balance of T cell subsets meaning fewer defensive regulatory T cells & more pro-inflammatory Th1/Th17 cells.
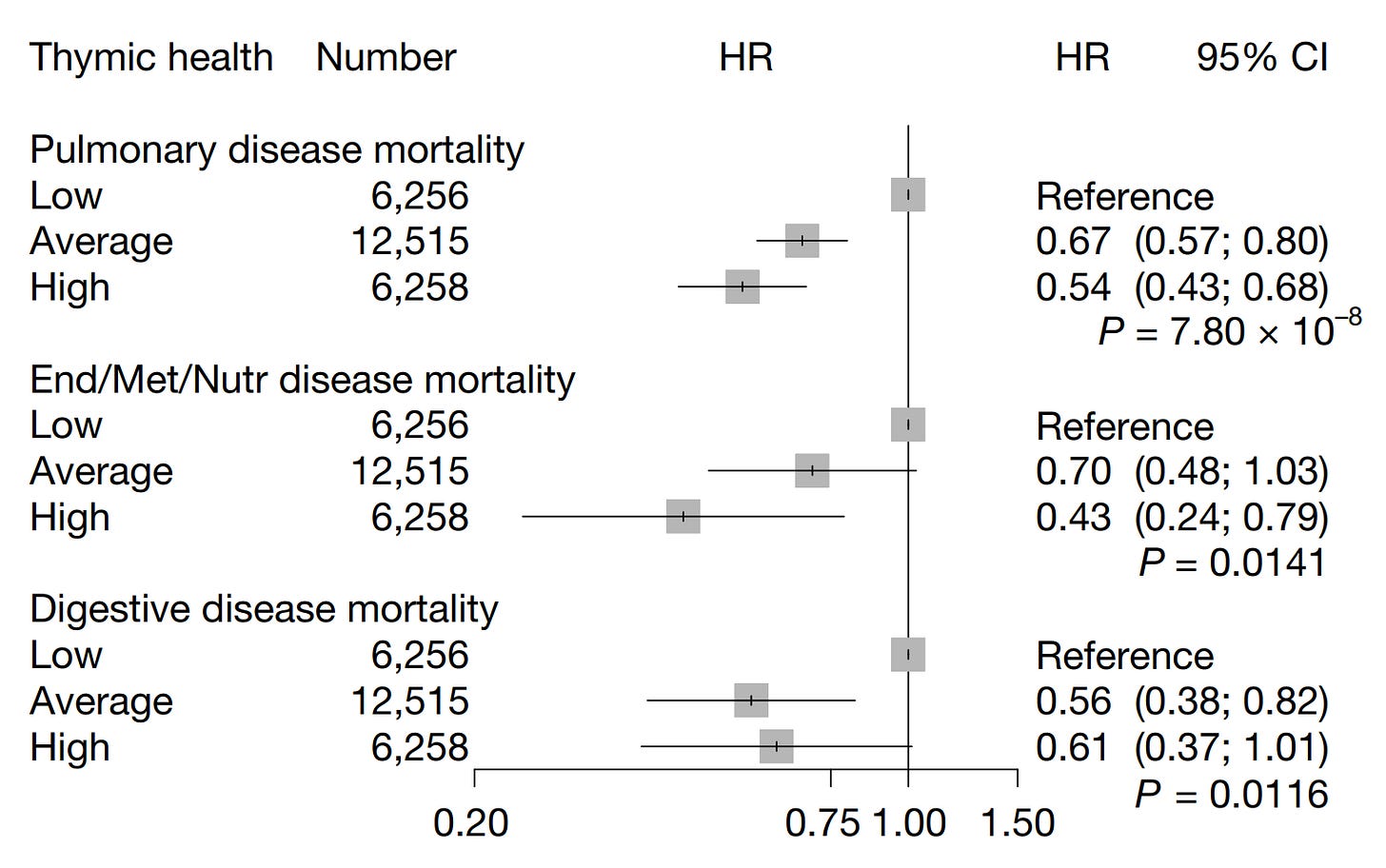
4. The effect extended across most disease categories.
Beyond cancer & CVD, those with higher thymic health were less likely to die from:
Pulmonary disease mortality: 61% lower for high thymic health
Endocrine, metabolic, & nutritional disease mortality: 68% lower
Digestive disease mortality (liver, gallbladder, pancreas): 54% lower
Thomas et al. (2020) showed that thymic involution has a dual-pathway model driving both immunosenescence (reduced immune competence) & inflammaging (chronic sterile inflammation).
T cells do more than fight infections. They recognize & eliminate senescent cells. So when thymic output drops, this clearance function fails, & the aging cell burden grows across every tissue.
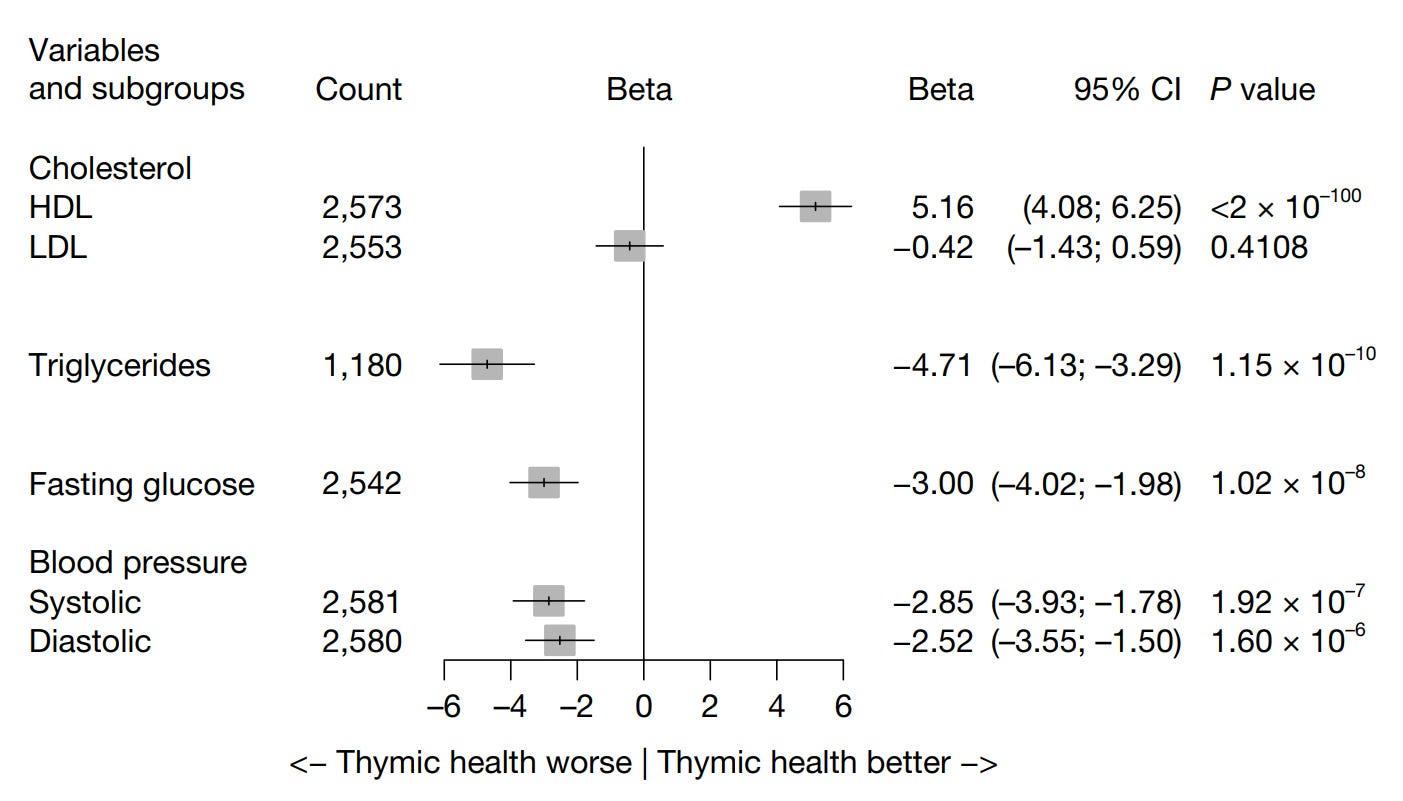
5. Chronic inflammation and metabolic dysfunction tracked with thymic decay.
When researchers analyzed blood inflammatory proteins in the FHS, 16 of 68 tested proteins were negatively correlated with thymic health. The list included IL-6, IL-18, VEGFA, HGF, and several CXCL chemokines, which are all major drivers of systemic inflammation, atherosclerosis, & cancer progression.
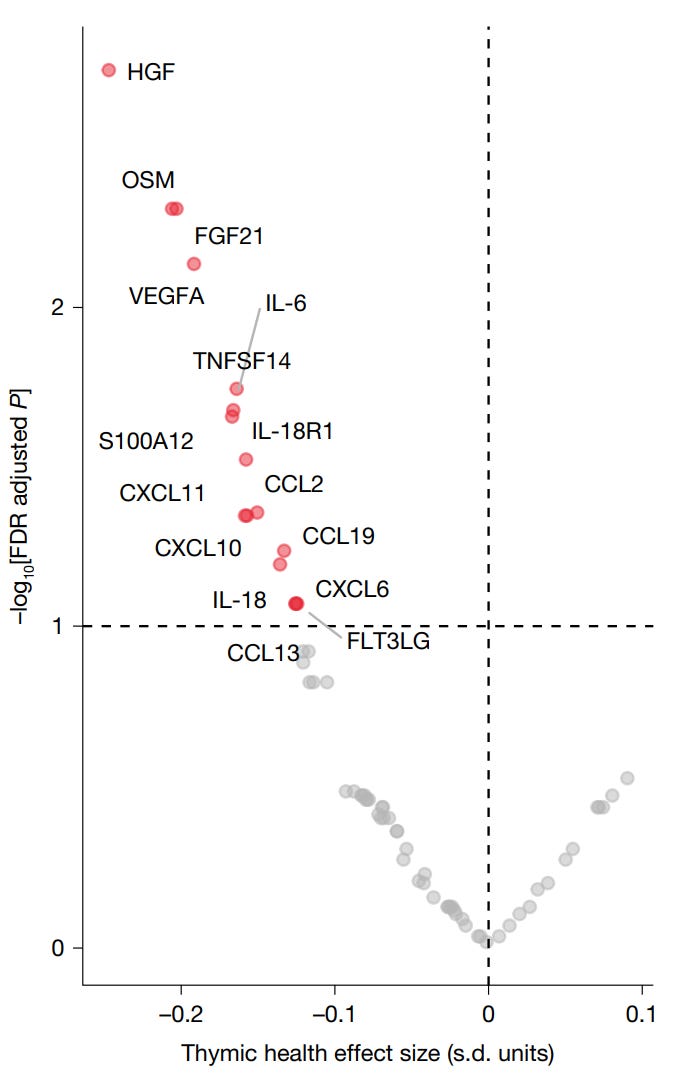
The same trends held true for cardiometabolic markers. Higher HDL cholesterol was positively correlated with better thymic health. Triglycerides, fasting glucose, & blood pressure were all negatively associated.
Another reason to keep a close eye on the trig/HDL ratio over time.
It shouldn’t come as a surprise, but obesity accelerates thymic involution (especially visceral fat tissue):
More visceral fat (inflammatory “organ”) → increased chronic inflammation accelerates thymic decay → thymic decay reduces immune competence → allows aging cell accumulation → senescent cells produce more inflammation → cycle compounds with age.
Promising Thymus Regeneration Paths
In September 2023, ARPA-H announced funding for a dedicated Thymus Rejuvenation program.
But the private sector is moving faster as multiple startups are now directly targeting thymus regeneration:
Intervene Immune → Brooke & Fahy’s company is furthest along clinically. Their TRIIM-X trial uses personalized combos of growth hormone, DHEA, & metformin to regrow thymic tissue in adults using already-approved drugs. So far interim results look promising.
Thymmune → Engineering functional thymus tissue from stem cells building replacements rather than regenerating what’s left.
TECregen → Emerged with $12.6M in seed funding to selectively regenerate thymic tissue using ligand engineering without the systemic toxicity that’s plagued growth factor approaches.
Tolerance Bio → $20.2M funded. Targeting immune tolerance through the thymus. Focused on autoimmune disease rather than pure regeneration.
Repair Biotechnologies → Developing gene & protein therapies to reverse thymic atrophy directly.
Where We Go From Here
An obvious question remains: “What can we do about it now?”
First, bioindividuality will remain key in the advancement of the therapeutic efforts moving forward. One of the best voices in our space summarized this:
And Dr. Bakri isn’t the only one paying attention:
We’ve had decades of asking why cancer patients fail immunotherapy, while the better question may have been whether their immune system was ever equipped for the fight to begin with.
Expect at least 1-2 regeneration protocols developed in 2026. As always, we’ll stay tuned & continue our research efforts on what promising regeneration protocols might look like in the future. More to come.
See you guys next week,
Phys











Any blood markers to test for thymus function?
What about running tb 500 if you think you have a poorly functioning thymus to mimick its effects?
Great post, BTPhys. Really glad to see the thymus hitting the limelight a little bit recently. Always been interested in the reduced cancer aspect most, since of the main killers, that's the one that trends closest to "crapshoot."
Will say, I read the Fahy TRIM paper with interest, but it in effect amounts to treatment with Growth Hormone... which I find pretty underwhelming. Will also note there were only 9 participants. Have never really seen research show a real mortality benefit or longevity benefits outside of weight/fat% to Growth Hormone treatment. The metformin and even the DHEA were more or less there to handle the GH side effects. Hopefully their upcoming larger + control group trial clears this up more, but I just remain skeptical that hitting people with GH and hoping it helps grow the thymus a bit is the answer.
That said, some of the other approaches - thanks for the links!! - look really interesting and promising and far more thymus targeted. To me, that's where the money will be.