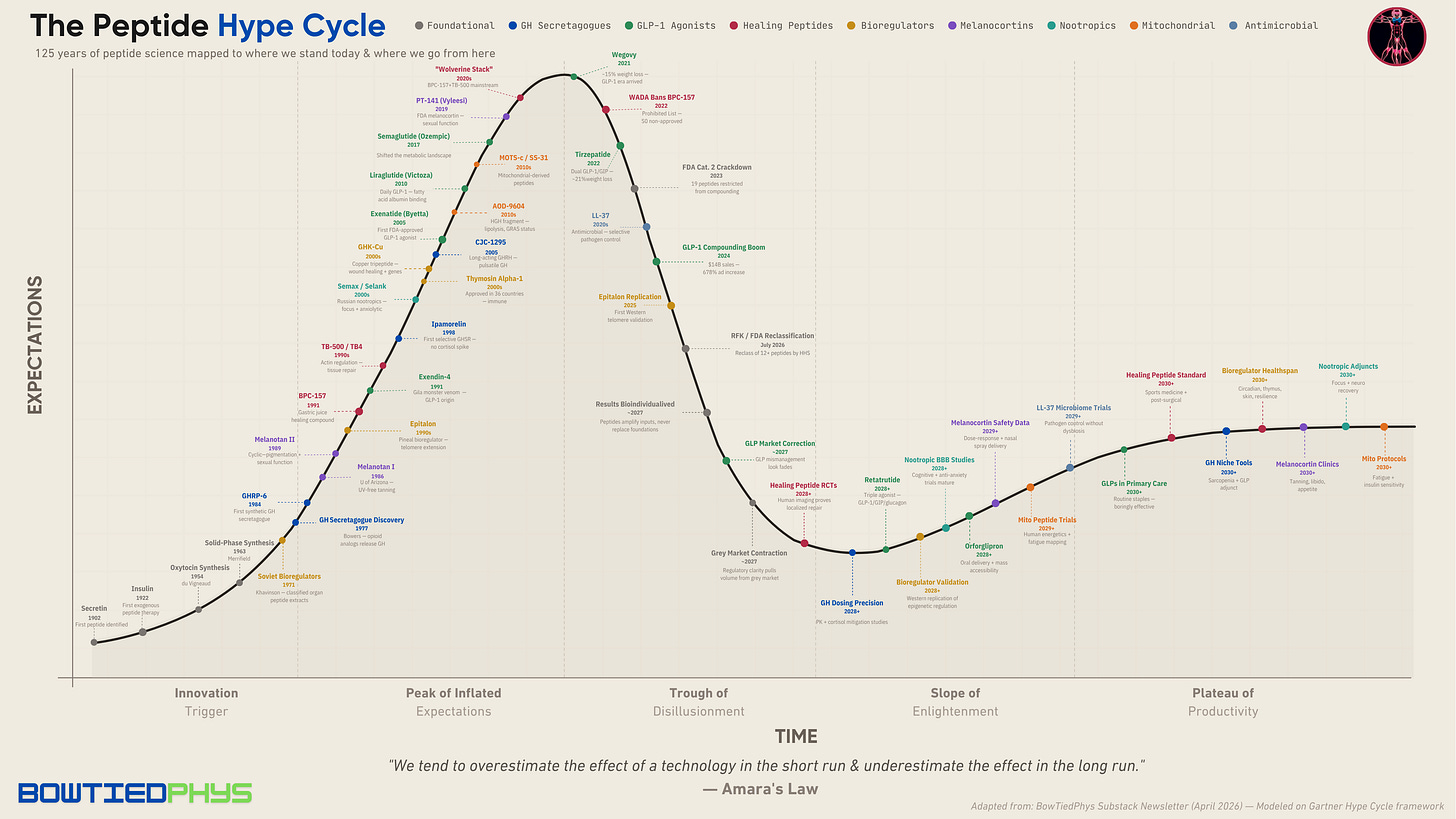
You're Late to a 125-Year-Old Peptide Party
The peptide timeline nobody has written until now & where we go from here
Ask someone about the origins of AI as we know it today and they’ll probably trace it back to 2022 when ChatGPT launched and became the fastest consumer app to hit 100M users in just 2 months. But few know the story actually started in 1956 at Dartmouth. It felt like it blossomed out of thin air, but it was really just building for more than seven decades.
Peptides follow the same arc – Amara’s Law:
“We tend to overestimate the effect of a technology in the short run & underestimate the effect in the long run.”
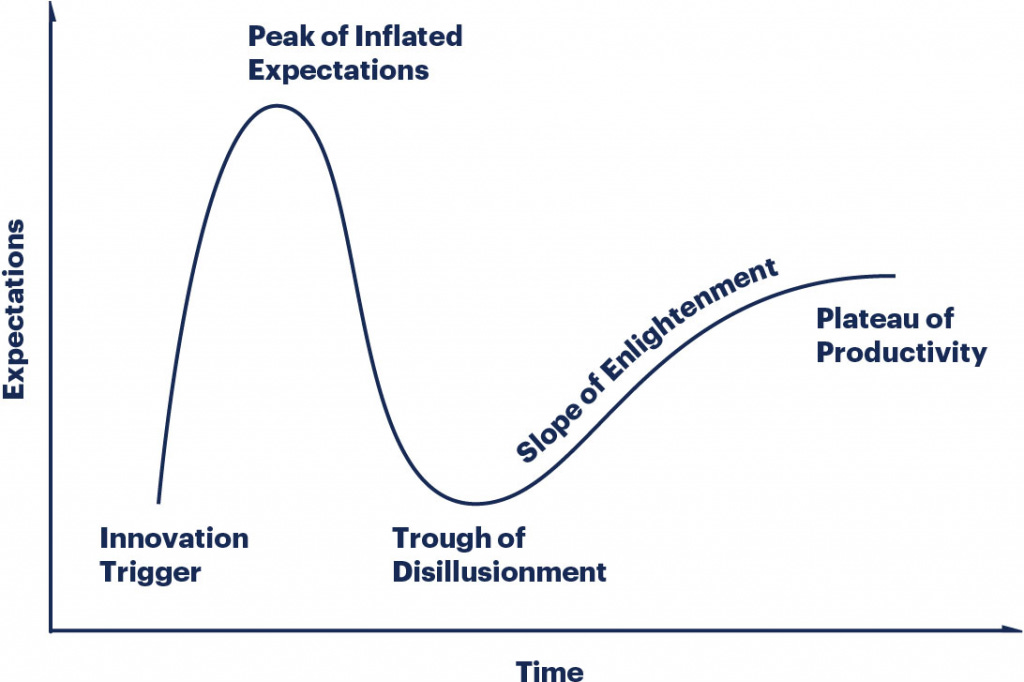
This idea is best modeled by the Gartner Hype cycle.
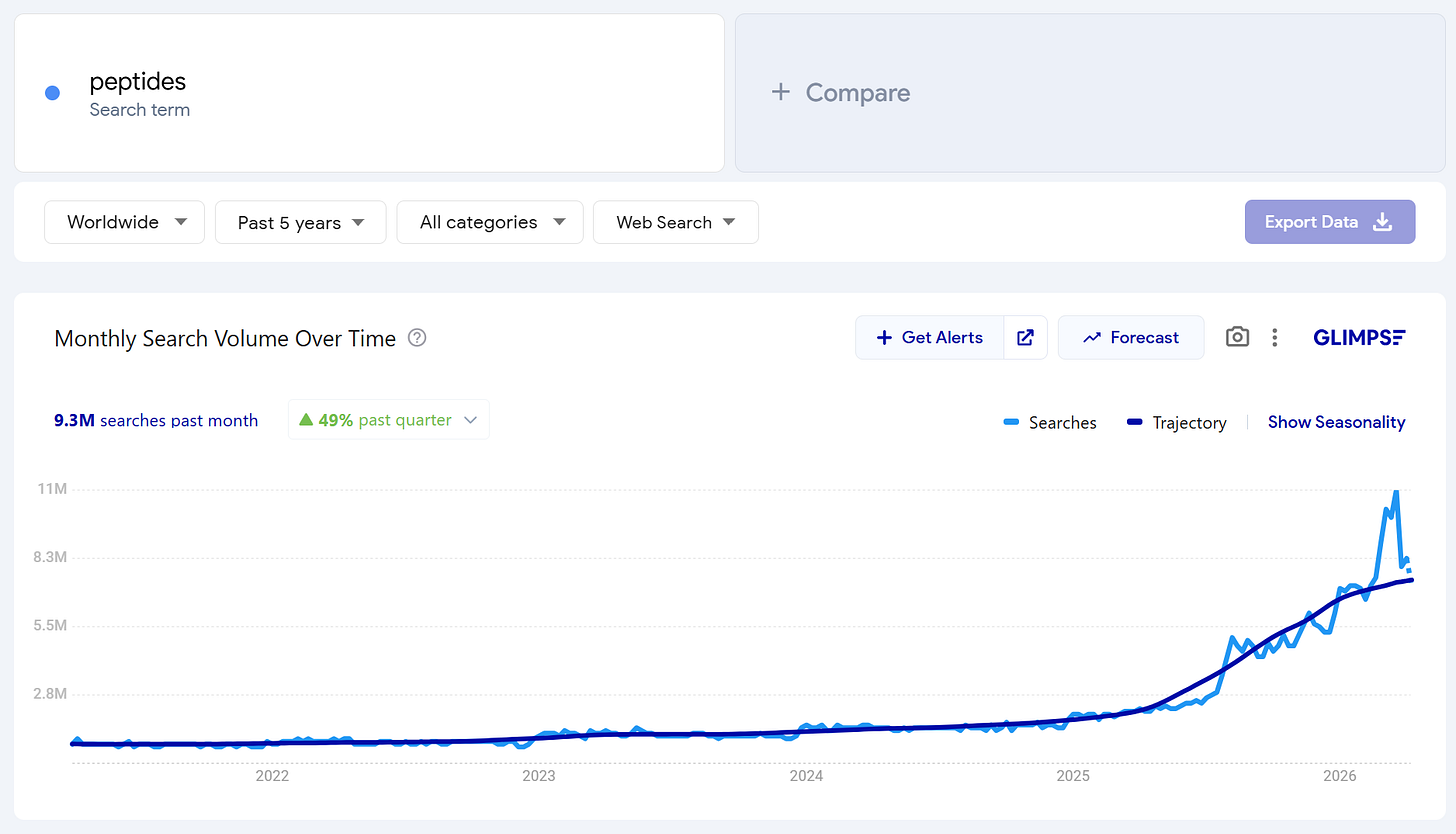
After a few mainstream podcasts and wave of viral X posts in 2025, “peptides” became the buzzword that means everything and nothing at the same time. To most, it all became relevant overnight.
But this short amino acid chain science has been building for over a century. Albeit quietly. In Soviet military labs, University of Arizona basements, Gila monster saliva, & Croatian gastroenterology departments.
As we write this in April 2026, we’re sitting near the peak of inflated expectations. And a sharp bifurcation is forming. The hype riders vs. the elite practitioners. The former as tactic hunters. The latter as strategic health infrastructure builders. If you’re reading this — congrats on finding your way into the latter.
Downloadable PDF plot below for detailed viewing (abridged version):
1902–1953: The birth of peptides
The first peptide ever identified came in 1902 when William Bayliss & Ernest Starling discovered secretin, a chemical messenger released from the gut lining signaling the pancreas to secrete digestive enzymes.
Two decades later in 1921, Frederick Banting & Charles Best isolated insulin from canine pancreases. By 1922, a dying 14-year-old diabetic named Leonard Thompson received the first human injection.
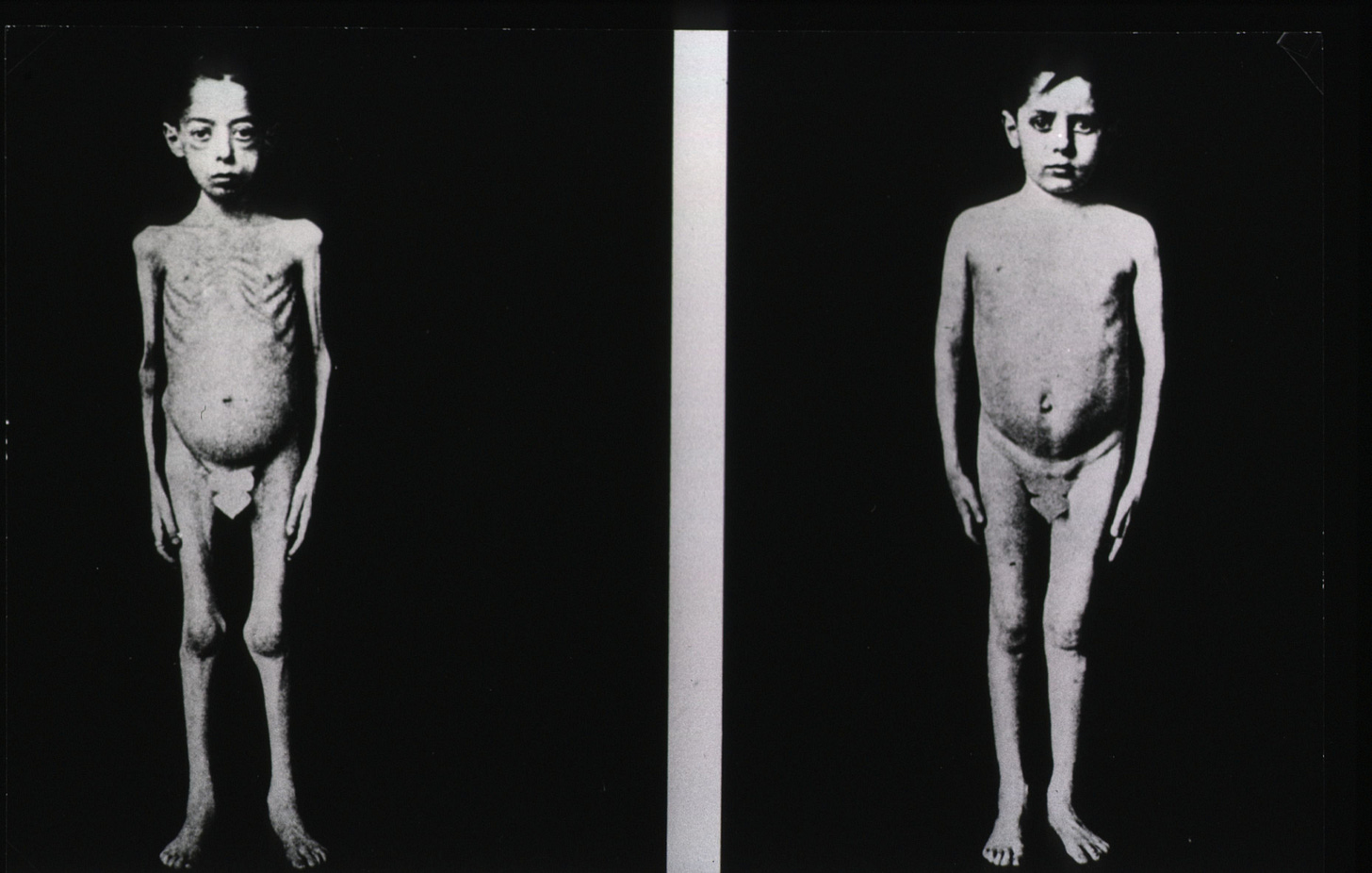
This marked the first exogenous peptide therapy ever used in humans. Insulin proved the model that every peptide discussed here follows: isolate or synthesize a signaling molecule → deliver it exogenously → restore or redirect a biological process.
In 1954, chemist Vincent du Vigneaud rang in the era of synthetic peptide design as we know it today. He completed the first total synthesis of oxytocin & vasopressin assembling peptides amino acid by amino acid proving they can be built from scratch. He won the Nobel Prize the following year.
1963–1979: The infrastructure gets built
Prior to 1963, making peptides in a lab was an arduous process. Every amino acid had to be coupled individually in solution, purified, & verified. Bruce Merrifield came along and invented solid-phase peptide synthesis (SPPS) that won him the Nobel Prize in Chemistry in 1984. SPPS automated the process by anchoring the growing chain to an insoluble resin bead making peptide production scalable.

Without SPPS, no peptide sold today would exist. Meanwhile, behind closed doors in the USSR, a mastermind was developing a parallel peptide vector.
Bioregulators: The classified Soviet program
In 1971, a military physician named Vladimir Khavinson started extracting peptide complexes from animal organs at the Kirov Military Medical Academy in Leningrad. The classified program objective was to protect soldiers, cosmonauts, & athletes from radiation exposure, extreme cold, & operational stress.
Khavinson’s team isolated short-chain peptides (2 to 4 amino acids long) from the thymus, pineal gland, brain cortex, prostate, & retina of calves. These became the first peptide bioregulators:
Thymalin (from the thymus) regulated cellular immunity.
Epithalamin (from the pineal gland) restored melatonin production and endocrine function.
Khavinson eventually proposed these ultra-short peptides could bind directly to DNA promoter regions and modulate gene expression. These were epigenetic switches that told aging cells to behave younger.
By 1982, Thymalin received formal registration for medical use in the USSR. Six peptide pharmaceuticals would eventually be approved for clinical use.
It wouldn’t be for another decade until the rest of the world heard of bioregulators.

Growth hormone secretagogues: An accidental discovery
In 1977, endocrinologist Cyril Bowers at Tulane University was studying small opioid-like peptides (enkephalin analogs) when he noticed certain chemical modifications that caused pituitary cells to release growth hormone.
This was the birth of the growth hormone secretagogue field.
1980–1989: Three parallel threads
The 1980s saw three separate research programs develop in parallel.
Thread 1: GH secretagogues mature
By 1984, Bowers synthesized GHRP-6 making it the first synthetic hexapeptide to dose-dependently release growth hormone. He was building the key before anyone knew the lock existed, which became known as “reversed pharmacology”. GHRP-6 worked through a receptor nobody had identified yet.
The endogenous hypothalamic hormone GHRH was discovered shortly after. Interest in GHRPs faded as most assumed GHRH was the primary GH regulator. That assumption turned out to be wrong. GHRP-6 & GHRH operated through completely distinct receptors and had synergistic effects on GH release.
Sermorelin, the synthetic form of GHRH, entered development during this period and would eventually receive FDA approval in 1997 as a diagnostic agent for GH deficiency.
Thread 2: Melanocortins emerge in the desert
At the University of Arizona, biologist Mac Hadley & chemist Victor Hruby were modifying alpha-melanocyte-stimulating hormone (α-MSH) — a naturally occurring peptide involved in skin pigmentation. Their goal was to create a stable synthetic analog that could induce tanning without UV exposure helping to reduce skin cancer risk.
Melanotan I came first: a linear 13-amino-acid peptide that closely resembled natural α-MSH but lasted longer in the body.
Then came Melanotan II: a cyclic heptapeptide that was much more potent and metabolically stable. Most peptides are straight chains where enzymes can attack the exposed ends and break them down fast. MTII had no free ends for enzymes to grab onto giving it longer lasting effects.
MT-II also bound to more than just MC1R (pigmentation) but also MC3R, MC4R, and MC5R. All of which were receptors involved in sexual function, appetite, & energy balance.
Thread 3: Soviet bioregulators go clinical
By the mid-1980s, Khavinson extracted over 20 peptide complexes from animal organs and synthesized 15 additional short peptides. Thymalin and Epithalamin had been in clinical use within the Soviet military medical service. Others followed: Cortexin (brain cortex, for cerebral function), Prostatilen (prostate), Retinalamin (retina).
Bioregulator treatment slowed age-related biomarker decline and suppressed tumor development in animal models showing lifespan increases of 20-40%.
But the work was still classified & published in Russian journals leaving it entirely invisible to Western science until the Soviet Union collapsed.
1990–1999: The discovery decade
Nearly every major category of peptide discussed online today has its roots in the 1990s.
BPC-157: Gastric juice yields a healing compound
In the early 1990s, Croatian pharmacologist Predrag Sikirić based at the University of Zagreb isolated a stable pentadecapeptide (15 amino acids) from human gastric juice. They named it Body Protection Compound-157 (BPC-157).
Sikirić sought to understand why the stomach heals its own lining despite constant exposure to hydrochloric acid. His team found a peptide fragment from a larger protective protein having cytoprotective effects across many tissue types including the gut, tendons, ligaments, muscle, bone, & vasculature.
Over the next three decades, Sikirić’s lab would produce the vast majority of published BPC-157 research: acceleration of wound healing, reductions in inflammation, & protection of GI lining from previous damage.
Exendin-4: The Gila monster connection
The Ozempic story is most unique in its origins. In 1991, endocrinologist John Eng was studying the venom of the Gila monster, a venomous lizard native to the American Southwest, when he identified a 39-amino-acid peptide called Exendin-4. It had extensive structural homology to human GLP-1, the incretin hormone that stimulates insulin secretion after meals.

The big difference between endogenous GLP-1 & Exendin-4: the former has a plasma half-life of ~2 min & Exendin-4 has a natural mutation extending its half-life to 2.5 hours.
After struggling to find institutional support, Eng patented Exendin-4 personally. He partnered with Amylin Pharmaceuticals years later to develop what became the first GLP-1 receptor agonist: exenatide.
Retrospectively, this was the start of a metabolic revolution. In 1991, the response was silence.
The Soviet collapse and bioregulator declassification
When the Soviet Union collapsed in 1991, Khavinson’s classified research became accessible. The St. Petersburg Institute of Bioregulation and Gerontology was established. The peptide bioregulator program transitioned from military secrecy to an open scientific publication.

Khavinson refined his approach as he shifted from crude organ extracts (Cytomaxes) to chemically synthesized short peptides (Cytogens). Epitalon became the most internationally recognized molecule from this program and one becoming increasingly popular today. In 2025, a UK-based group published the first independent replication confirming dose-dependent telomere extension in normal human cells
The synthetic bioregulator Thymogen (a dipeptide for immune correction) also emerged from this era. By the 2000s, Khavinson amassed over 105 patents and 775 published papers spanning four decades of research.
GH secretagogues: 2nd generation
The GHRP family expanded rapidly through the 1990s.
GHRP-2 (pralmorelin) was synthesized as a more potent second-generation analog having a stronger GH release than GHRP-6 with better sustained elevation over time. Hexarelin followed with similar potency.
In 1998, Novo Nordisk characterized ipamorelin as the first selective growth hormone secretagogue. Unlike GHRP-6 and GHRP-2, ipamorelin released GH without spiking cortisol, ACTH, or prolactin.
Melanotan II: Too hard to ignore (*winks*)
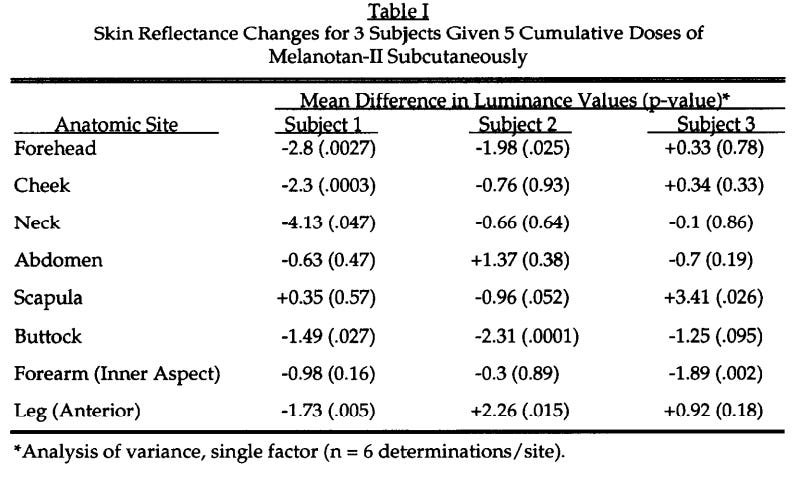
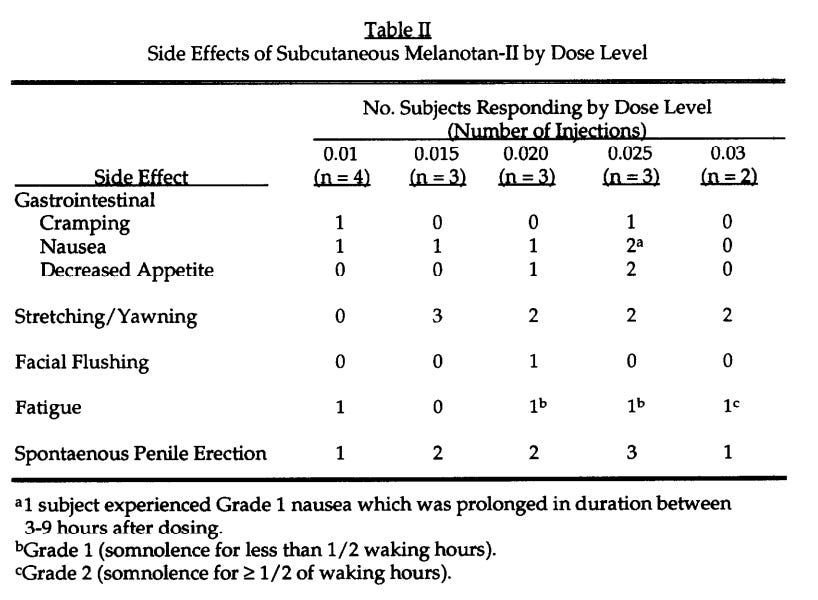
In 1996, the Arizona team ran a Phase I clinical trial of Melanotan II to evaluate its tanning efficacy. Five low-dose subcutaneous injections produced visible skin darkening. The tanning worked.
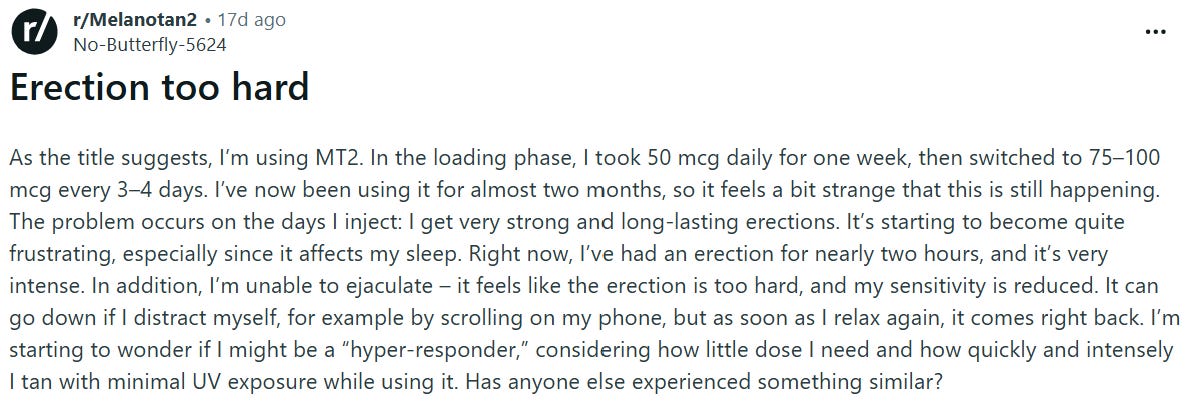
But MT-II’s broader receptor binding demonstrated unforeseen outcomes…inducing erections in each male subject. MC4R activation in the central nervous system was triggering penile erection through hypothalamic / spinal pathways independent of sexual arousal.
Many of the forums & Reddit threads have been detailing these very cases over the last decade.
Despite this not being the intended objective, it redirected the entire melanocortin research program. Sexual dysfunction became the new target. PT-141 (bremelanotide) was synthesized by Palatin Technologies as a derivative of the MT-II scaffold, which optimized for central melanocortin activity with reduced melanogenic effects.
Thymosin Beta-4 & TB-500
Allan Goldstein first described Thymosin Beta-4 (TB4) in the 1960s, but it didn’t gain traction until the 1990s. TB4 functions by maintaining a ready-to-deploy reservoir that cells can rapidly assemble into structural filaments when tissue is damaged. This is what allows reparative cells like fibroblasts, stem cells, & endothelial cells to physically migrate into a wound site and start rebuilding.
The synthetic fragment TB-500 was a TB4 fork, which distilled it into a shorter, more accessible molecule. It’s now become one of the most widely used recovery peptides in sports performance & recovery alongside BPC-157.
2000–2009: From labs to prescription pads
GLP-1 goes pharmaceutical
After a 14-yr period, the FDA approved the Gila monster peptide (exenatide (Byetta)) as the first GLP-1 receptor agonist for type 2 diabetes in 2005.
Novo Nordisk watched closely and began developing its own GLP-1 analog. Liraglutide used a different strategy. They attached a fatty acid chain (C16) to the GLP-1 peptide, allowing it to bind reversibly to albumin in the bloodstream. This slowed renal clearance and extended the half-life enough for a single daily dosing. It received EU approval in 2009 and went on the market as Victoza.
CJC-1295: Long-acting GHRH
ConjuChem Biotechnologies developed CJC-1295, a modified version of GHRH(1-29) with a Drug Affinity Complex (DAC) that bound to serum albumin extending its half-life to 6-8 days. The “no-DAC” version (Mod GRF 1-29) used the same GHRH analog without the albumin-binding complex. It was shorter acting requiring more frequent dosing, but it preserved pulsatile GH release that mirrors natural endocrine rhythms. Phase II trials showed dose-dependent GH increases of 2-10x sustained for 6+ days after a single injection.
Khavinson publishes landmark human data
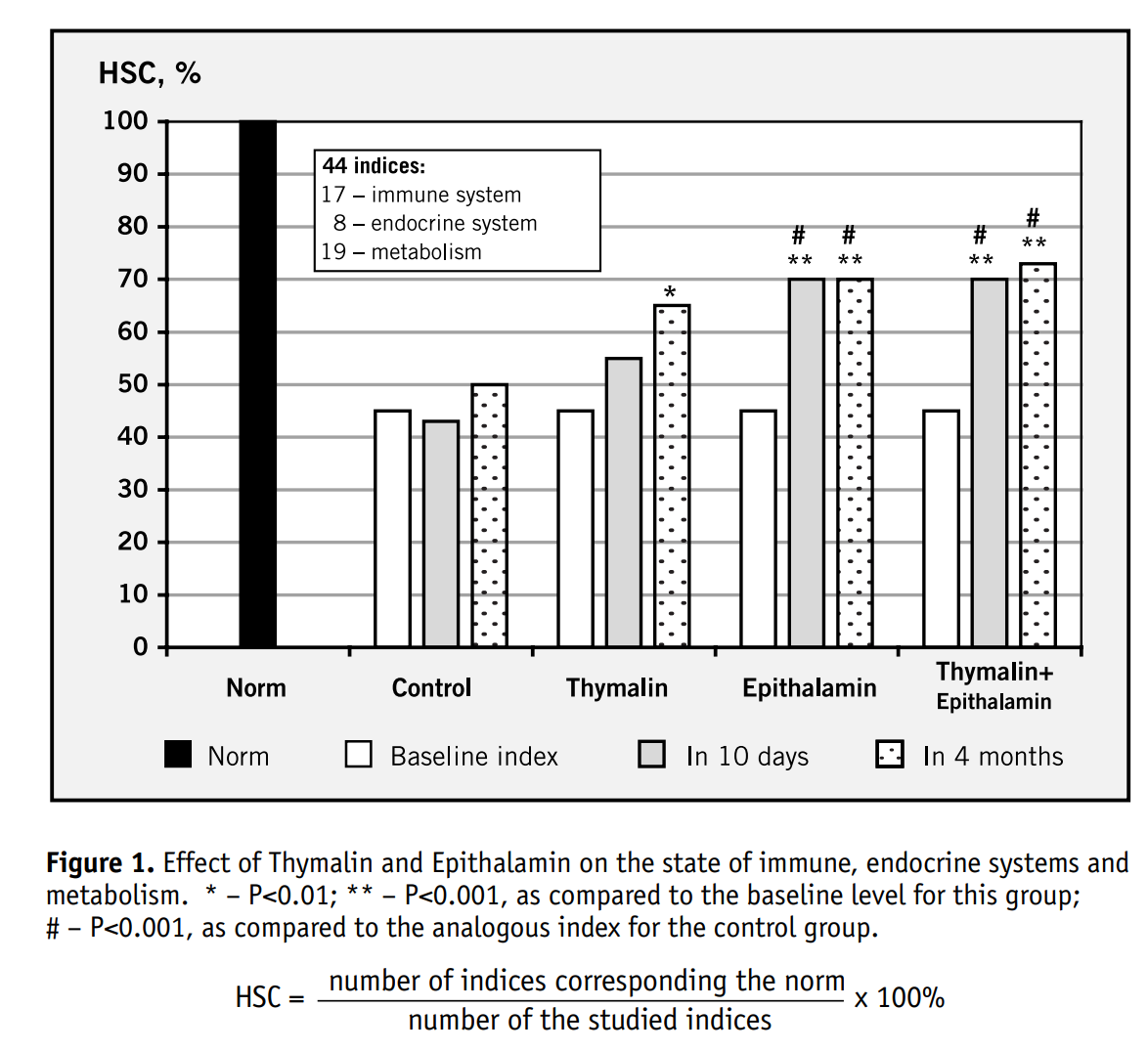
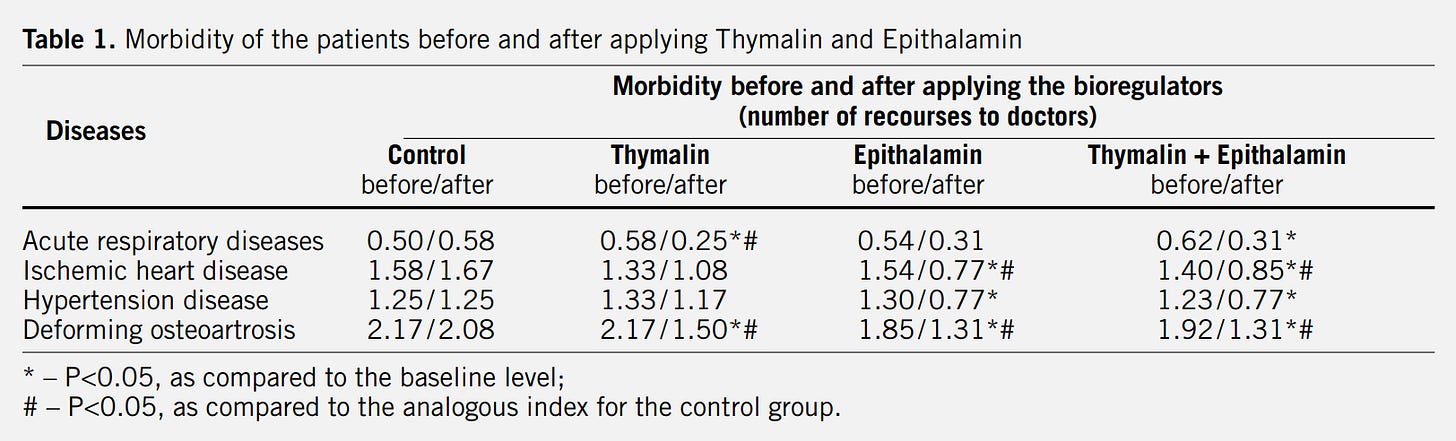
In 2003, Khavinson & Morozov published the results of a 6-8 year clinical assessment of Thymalin & Epithalamin in 266 elderly subjects. The bioregulators normalized immune function, endocrine markers, & cardiovascular indices.
Mortality in the treatment groups was 28% lower than controls.
Though the story was compelling, the data came from Russian institutions and was published in Russian journals, which was met with Western skepticism.
Thymosin Alpha-1: Approved everywhere but the US
Thymosin Alpha-1 (Ta1) is a 28-amino-acid peptide derived from thymus tissue. It was approved under the brand name Zadaxin in 36 countries for hepatitis B, hepatitis C, & as an immunotherapy adjunct, though it’s never been submitted for FDA approval in the US.
Nootropic peptides: Selank and Semax
Two synthetic peptides with nootropic & anxiolytic properties emerged from Russian pharmaceutical development.
Semax: a synthetic analog of ACTH(4-10) with a Pro-Gly-Pro tripeptide extension known for its neuroprotection and was approved in Russia for anti-stress, cognitive enhancement, stroke recovery, & ADHD.
Selank: a synthetic analog of the immunomodulatory peptide tuftsin, also with a Pro-Gly-Pro extension showing anxiolytic effects (comparable to benzodiazepines without side effects/dependency risk) and was approved in Russia for generalized anxiety.
GHK-Cu: A copper tripeptide
In 1973, biochemist Loren Pickart discovered GHK-Cu (glycyl-L-histidyl-L-lysine complexed with copper) as a naturally occurring human tripeptide first identified in human plasma.
Endogenous concentrations decline significantly with age → plasma levels at age 60 (~80 ng/mL) are less than half at age 20 (~200 ng/mL).
GHK has now become a staple in the cosmetic & regenerative health space either applied topically or applied subcutaneously.
Through the 2000s, its research profile widened considerably given it modulates expression of hundreds of genes tied to:
wound healing
collagen synthesis
antioxidant defense
extracellular matrix remodeling
Mechanistically, GHK-Cu is unique in that it acts like a bioregulator but drives direct tissue repair like a healing peptide. And that’s why we’re seeing it show up in so many different protocols.
2010–2019: The pharmaceutical explosion
GLP-1 becomes a metabolic optimization story
The 2010s marked the shift of GLP agonists from a niche T2 diabetes treatment to one of the most powerful metabolic optimization tools in existence.
After liraglutide’s (Victoza) 2010 FDA approval in 2010 for type 2 diabetes, Saxenda (liraglutide at higher doses) became the first GLP-1 approved specifically for weight loss.
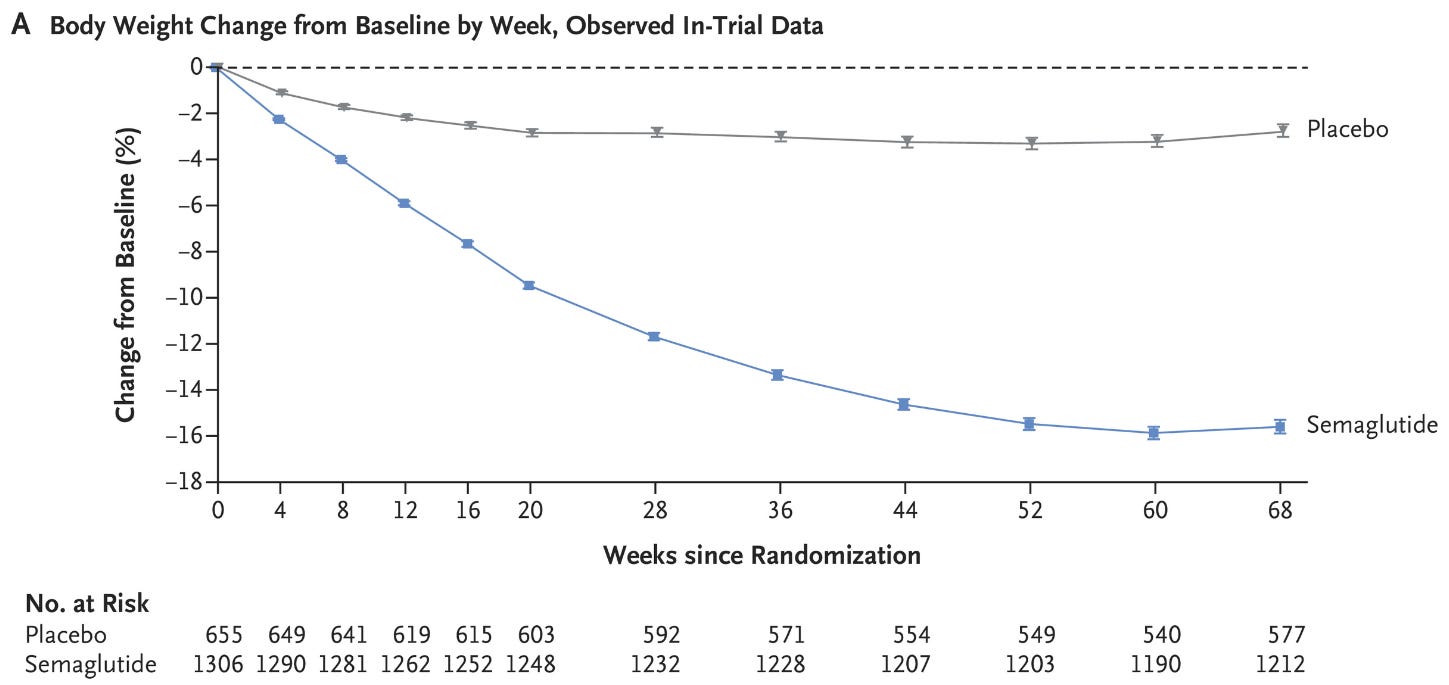
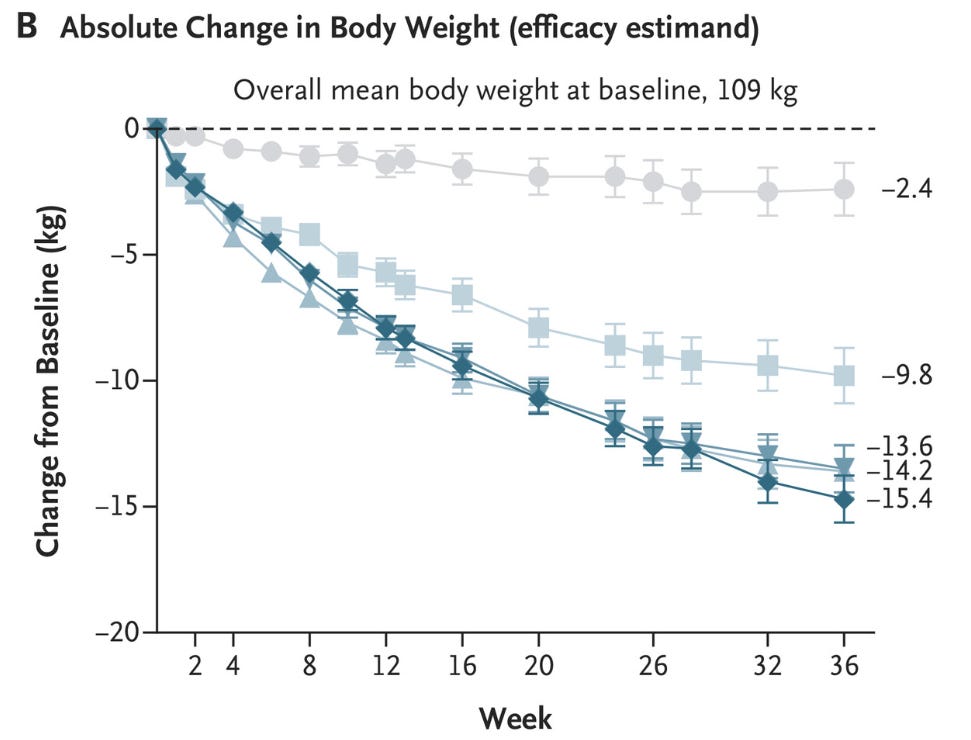
Shortly after, dulaglutide (Trulicity) arrived with once-weekly dosing. Then in 2017, semaglutide (Ozempic) shifted the entire metabolic landscape.
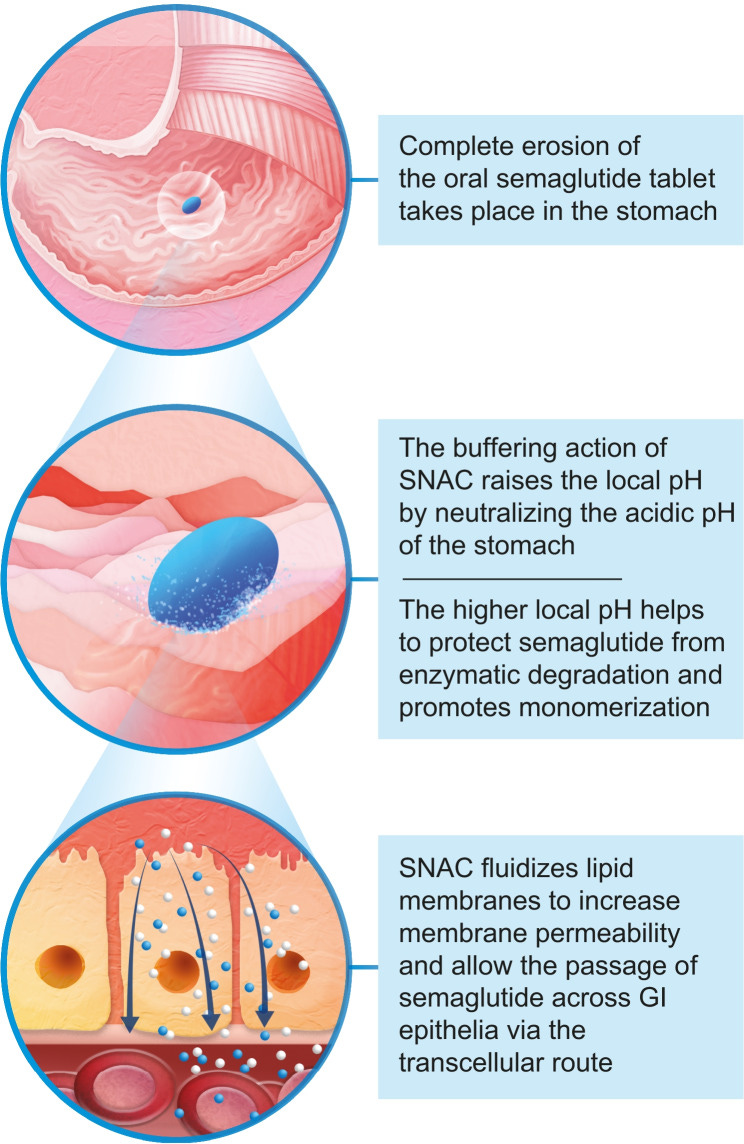
By 2019, oral semaglutide (Rybelsus) proved that a peptide could survive stomach acid and reach therapeutic levels orally. This was done via a co-formulated absorption enhancer called SNAC that raised local stomach pH and improved absorption.
Melanocortins reach the market
In 2014, the EU approved Melanotan I (Scenesse) for a rare condition causing extreme photosensitivity. This marked the first regulatory validation of melanocortin-based pigmentation therapy. The US followed with FDA approval in 2019.
That same year, bremelanotide (PT-141) sold under the brand name Vyleesi received FDA approval for hypoactive sexual desire disorder in premenopausal women.
Melanotan II has never received regulatory approval though. Its non-selective receptor binding (what made it versatile) also caused some regulatory complications.
The underground peptide economy gains momentum
By the late 2010s, a grey market peptide economy matured. Compounding pharmacies were focused on formulating BPC-157, TB-500, ipamorelin, & CJC-1295 for clinical use under physician prescription. The “Wolverine Stack” (BPC-157 + TB-500 for accelerated tissue repair) entered the world of sports medicine & decentralized biohacking communities.

Mitochondrial peptides are born
A newer class of peptides derived from mitochondrial DNA began attracting research attention.
MOTS-c: a mitochondria-derived peptide known for its effects on metabolic regulation & insulin sensitivity.
Humanin: another mitochondria-derived peptide known for its neuroprotective properties in Alzheimer’s disease models & anti-apoptotic effects.
AOD-9604: a modified fragment of HGH developed specifically for its fat-reducing properties without the diabetogenic effects of full-length HGH. It received Generally Recognized as Safe (GRAS) status from the FDA as a food ingredient.
2020–2026: Peak hype + peak friction
The GLP era
In 2021, Wegovy (semaglutide) received FDA approval for chronic weight management.
Average weight loss in clinical trials = ~15% of body weight. The GLP-1 era officially arrived.
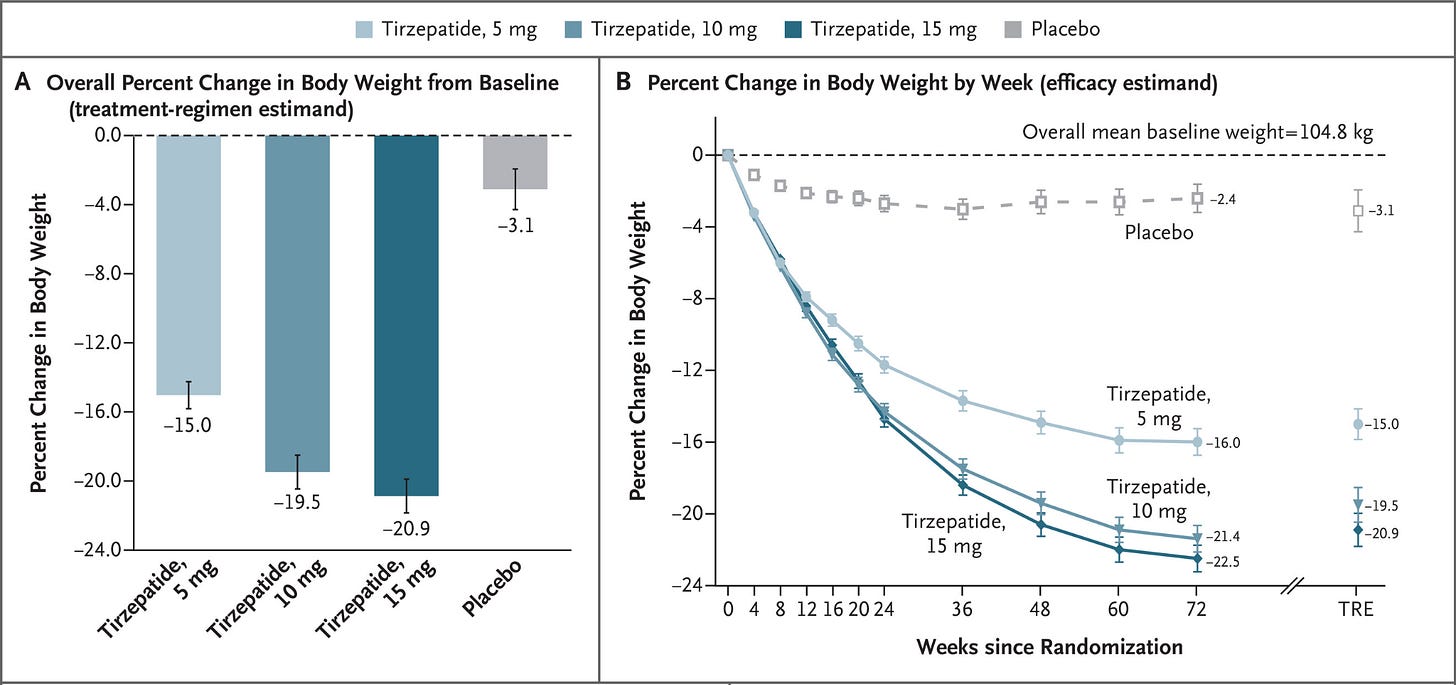
In 2022, tirzepatide (Mounjaro) received FDA approval as the first dual GLP-1/GIP agonist for T2 diabetes.
Clinical trials showed ~21% average weight loss.
In 2023, tirzepatide (Zepbound) received obesity approval. By 2024, GLP-1 drug sales neared $14 billion.
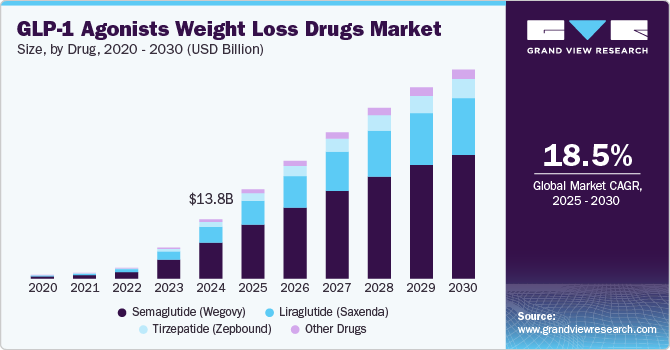
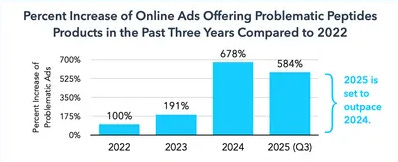
By the end of 2024, soaring demand brought on new shortages. Compounding pharmacies began producing semaglutide copies under shortage exemptions. Chinese peptide imports to the US doubled to $328 million. Online peptide advertising increased 678% from 2022.
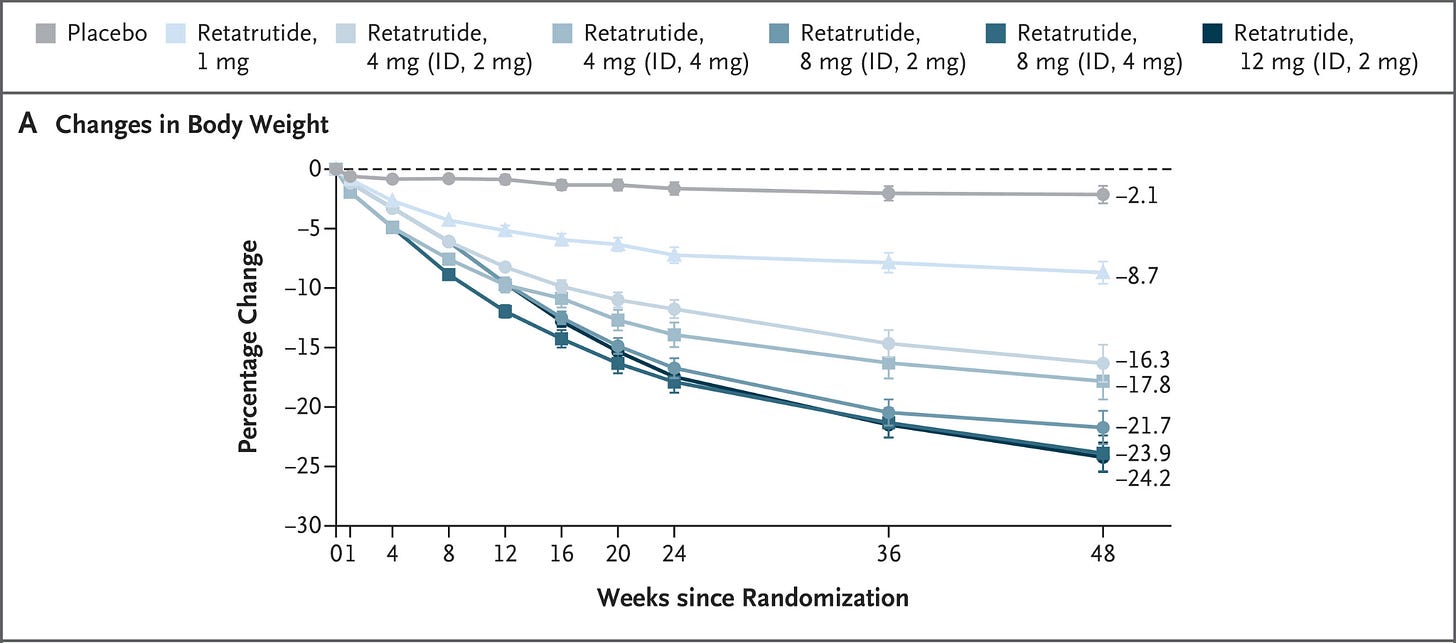
Meanwhile, the research pipeline continued moving. Retatrutide, a triple agonist targeting GLP-1, GIP, & glucagon receptors, entered late-stage clinical trials.
Orforglipron, an oral, non-peptide GLP-1 receptor agonist from Eli Lilly, advanced toward potential FDA approval. Though it remains much less effective than its injectable predecessors.
WADA says enough
In January 2022, the World Anti-Doping Agency added BPC-157 to its Prohibited List under category S0 (Non-Approved Substances). GHRPs (GHRP-2, GHRP-6, hexarelin, ipamorelin) were already banned under S2 (Peptide Hormones and Growth Factors).
FDA crackdown
In September 2023, the FDA placed 19 peptides in Category 2 meaning compounding pharmacies could no longer legally produce them. The stated rationale across these compounds included immunogenicity risk, peptide-related impurities, & insufficient human safety data.
Demand didn’t disappear. If anything, the restrictions amplified interest. Many turned to research chemical vendors and unregulated online suppliers. And so the peptide wild west was born.
Not without growing pains though. Enforcement escalated in June 2025 when the FDA raided Amino Asylum. More than 50 warning letters went to GLP-1 compounders a few months later and state-level enforcement followed. By March 2026, one of the biggest hubs, Peptide Sciences, voluntarily closed its doors.
The reclass
On February 27, 2026, HHS Secretary RFK, Jr. appeared on Joe Rogan hinting that he expects ~14 of the 19 previously restricted peptides to return to Category 1 status restoring the ability of licensed compounding pharmacies to produce them.
6 weeks later on April 15, 2026, the FDA made it official with a formal meeting scheduled for July 2026 where panelists will consider whether 12 peptides should be permitted for routine compounding.
2026 & beyond: Where we go from here
As of mid-2026, we’re sitting at the Peak of Inflated Expectations marked by the Hollywood GLP mismanagement look & the predatory TikTok affiliate culture.

Collectively, all of it has pushed expectations beyond what the evidence currently supports. And to be clear – the evidence is substantial by iteself.
So where do we go from here?
Like most technology curves, the correction is acute followed by a long-term stabilization.
The timeline forward is characterized by a few principles:
Real-world friction corrects the overshoot: Results are going to be bioindividualized & health context-dependent. Many will begin to discover that peptides amplify most lifestyle inputs rather than replace them. In other words, you can properly dose the MOTS-c all you want, but if diet isn’t dialed in, training is inconsistent, & sleep quality/circadian regulation is subpar, outcomes will fall short of expectations.
Maturing human data replaces hype with bioindividual precision: Expect to see much of Khavinson’s work replicated and even more mechanistic studies. Do not expect the RCTs to originate from the NIH, but instead from decentralized institutions/clinics running their own. Even the language shifts from generalized “peptides” to highly individualized, objective-driven interventions.
Oral / nasal delivery & commoditization strip away the mystique: The one thing standing in the way more than any other component for most is the fear of needles. A 2022 survey put that number at ~63% having at least some fear. Orforglipron-style molecules (oral + mass-manufacturable) transform GLP-1 therapy from an exclusive injectable to a widely accessible oral drug. This drives the most adoption and where we cross the chasm from early adopted crowd to gen pop adoption.
Regulatory stabilization prevents a deep trough: The RFK-led HHS reclassification (now formalized with a July 2026 FDA review) restores compounding access and pulls volume out of the gray market.
Beyond 2027, here’s what the Slope of Enlightenment & eventual Plateau of Productivity look like for each peptide category:
GLPs: On the Slope, more large-scale outcome trials and decentralized data gathering clarify metabolic effects while microdosing/adjacent protocols mitigate side effects & muscle loss. By the Plateau, they sit as routine staples integrated into primary care and boringly effective lifestyle adjuncts.
Healing peptides (BPC-157, TB-500/thymosins): The Slope brings targeted human RCTs and imaging data from decentralized, broscience style sources. This proves localized repair without systemic risks. On the Plateau, they become standard adjuncts in gut healing, sports medicine & post-surgical protocols.
GH secretagogues: Maturing pharmacokinetic and safety studies on the Slope refine dosing windows & cortisol mitigation. At the Plateau, they stabilize as niche tools for GH deficiency cases, muscle loss in older adults, & GLP adjacent protocols.
Bioregulators (Khavinson short peptides like Pinealon & Epitalon + GHK-Cu): Western replication trials on the Slope finally validate epigenetic gene regulation & oral bioavailability. By the Plateau, they emerge as foundational healthspan optimization tools for circadian rhythm regulation, skin, hair, thymus, & stress resilience.
Nootropic peptides (Selank, Semax): The Slope brings clean cognitive and anti-anxiety trials detailing blood-brain-barrier penetration and neurotransmitter modulation. On the Plateau, they settle as targeted adjuncts for focus, recovery from stress, or mild neuroinflammation especially in older adults with early onset neurodegeneration.
Melanocortins: Dose-response & long-term safety data on the Slope define targeted therapeutic windows for tanning, libido, & appetite with less of the old nausea worries. At the Plateau, they become staples at specialized med spas & peptide clinics. More accessibility, female adoption, & mobility enabled by nasal spray applications.
Antimicrobial peptides (LL-37): The Slope sees microbiome and infection outcome studies proving selective pathogen control without dysbiosis. The Plateau integrates these as next-generation adjuncts for gut, skin, and chronic low-grade infections.
Mitochondrial peptides (MOTS-c, SS-31): Early human energetics & fatigue trials on the Slope map vitality gains. By the Plateau, they anchor regenerative protocols for mitochondrial decline targeted for insulin/leptin sensitivity, chronic fatigue, or age-related vitality loss.
Final word
Before we depart, here’s one practical takeaway to remember: peptides amplify inputs & will never replace the lifestyle boulders.
If the first century of our peptide timeline built the molecules, the next decade will build the infrastructure of sovereign decentralized testing, cleaner clinical evidence, & delivery innovation.
We’re cautiously optimistic as we continue our own research & spread the lessons learned from the research of other elite practitioners in the community.
In the meantime, steer clear of Peptide TikTok, study what’s presented here (much more to come), & stay elite. We’ll see you guys next week.
Your friend,
Phys









Great article! But cerebrolysin is missing I'd say, it's a big one in peptide History I reckon